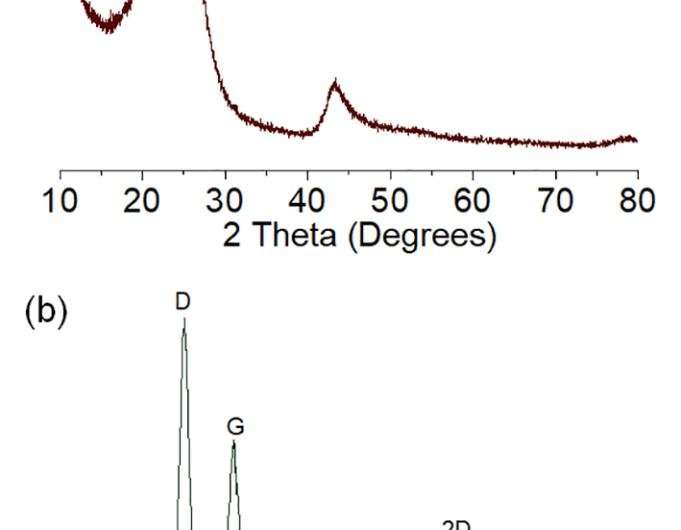
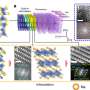
(a) XRD pattern of the as-prepared sample indexed with amorphous and graphitized peaks. (b) Raman spectra of D, G, D+D'', 2D, D+G and 2D' bands. Credit: Science China Press
Developing high-capacity carbon anode materials can further improve the energy density of sodium-ion batteries (NIBs). Recently, researchers from the Institute of Physics, Chinese Academy of Sciences (IOP-CAS), reported a high-capacity carbon anode (~400 mAh g-1) for NIBs. The results are published in Science Bulletin.
Since 2010, sodium-ion batteries (NIBs) have been intensively investigated because of their cost and resource advantages and possible applications in large-scale energy storage systems. However, the energy density of current NIBs remains a serious challenge, hindering large-scale commercial applications. Hard carbon is one of the most promising anodes in the early commercial NIBs for high capacity (~330 mAh g-1), good cycling stability, high initial Coulombic efficiency, reasonable cost, and the natural abundance of precursor materials.
Although extensive efforts have been devoted to the development of high-performance carbon anode materials, a consistent behavour in the discharge-charge curve is often presented with two distinct regions: a slope region above ~0.1 V and a plateau region below ~0.1 V. Usually, the plateau region exhibits a higher capacity than that of the slope region, and a high-capacity carbon anode often shows a large proportion of the plateau capacity, which could further increase the energy density of a full cell in terms of the average voltage to some extent. Therefore, designing and discovering a carbon anode with a large proportion of the plateau capacity can be a potential approach to increase the energy density of NIBs.
Recently, the team of Prof. Yong-Sheng Hu at the Institute of Physics, Chinese Academy of Sciences (IOP-CAS) reported a bi-honeycomb-like architecture carbon material by carbonizing a kind of charcoal in a high-temperature graphite furnace at 1900 °C. This carbon anode shows a high capacity of ~ 400 mAh g-1, which is higher than the ~ 330 mAh g-1 capacity of current hard carbon materials. About 85 percent (> 330 mAh g-1) of its total capacity is derived from the long low-potential plateau below ~ 0.1 V, which differs from the curves of typical hard carbon materials of NIBs. When coupled with air-stable Na0.9Cu0.22Fe0.30Mn0.48O2 layered cathode, a high energy density of ~240 Wh kg-1 was obtained with good rate capability and cycling stability. The discovery of this promising carbon anode is expected to draw further research toward high-energy-density NIBs for large-scale electrical energy storage.
-
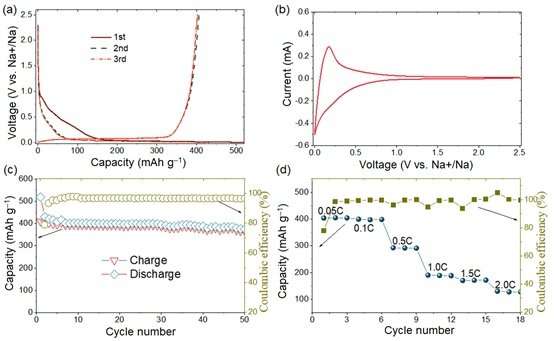
Electrochemical properties of this anode in half cells. (a) Galvanostatic discharge-charge curves, (b) CV curves, (c) Cycling performance and (d) Rate capability. Credit: Science China Press
-
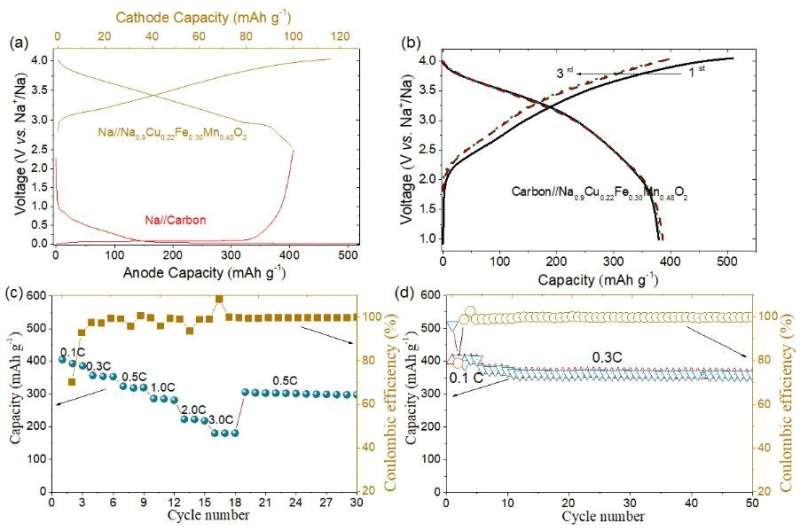
(a) Schematic illustration of charge-discharge voltage profiles of the carbon anode and Na0.9Cu0.22Fe0.30Mn0.48O2 cathode, respectively. Electrochemical performance of the carbon anode//Na0.9Cu0.22Fe0.30Mn0.48O2 the full cell, (b) Charge-discharge curves, (c) Rate capability and (d) Cycling performance. Credit: Science China Press
More information: Chenglong Zhao et al, High-temperature treatment induced carbon anode with ultrahigh Na storage capacity at low-voltage plateau, Science Bulletin (2018). DOI: 10.1016/j.scib.2018.07.018
Provided by Science China Press