Scientists find way to help fuel cells work better, stay clean in the cold

In a study to publish in Nature on January 31, researchers at the University of Science and Technology of China (USTC) report advances in the development of hydrogen fuel cells that could increase its application in vehicles, especially in extreme temperatures like cold winters.
Hydrogen is considered one of the most promising clean energy sources of the future. Hydrogen fuel cells have high energy conversion efficiency and zero emissions. But the development of hydrogen fuel cells faces many challenges, including the issue of carbon-monoxide (CO) damage of the fuel cell electrodes.
Currently, hydrogen is mainly derived from processes like steam reforming of hydrocarbons, such as methanol and natural gas, and water gas shift reaction. The resulting hydrogen usually contains 0.5 percent to 2 percent of trace CO. As the "heart" of hydrogen fuel cell vehicles, fuel cell electrodes are easily contaminated by CO gas impurity, resulting in reduced battery performance and shortened life, which severely hampers the application fuel cells in vehicles.
Earlier research has identified a method called preferential oxidation (PROX) as a promising way to remove trace amounts of CO from hydrogen by using catalysts. However, existing PROX catalysts can only work in high temperatures (above room temperature) and within a narrow temperature range, hindering practical applications such as fuel cell vehicles, which must be reliable even in winter months.
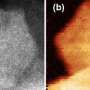
Now, a USTC team led by Junling Lu, professor at the Hefei National Laboratory for Physical Sciences at the Microscale, has designed a new structure of atomically dispersed iron hydroxide on platinum nanoparticles to purify hydrogen fuel over a broad temperature range of 198 to 380 degrees Kelvin. They also found that the material provided protection of fuel cells against CO poisoning during both frequent cold-starts and continuous operations in extremely cold temperatures.
"These findings might greatly accelerate the arrival of the hydrogen fuel cell vehicle era," said Prof. Lu. "Our ultimate goal is to develop a cost-effective catalyst with high activity and selectivity that provides continuous on-board fuel cell protection and that enables complete and 100 percent selective CO removal in a fuel cell that can be used for broader purposes."
One referee of the article commented: "When comparing with other catalyst systems reported in the literature, this reverse single-atom catalyst appears the best in terms of activity, selectivity, and stability in CO2-containing streams."
More information: Atomically dispersed iron hydroxide anchored on Pt for preferential oxidation of CO in H2. DOI: 10.1038/s41586-018-0869-5 , www.nature.com/articles/s41586-018-0869-5
Journal information: Nature
Provided by University of Science and Technology of China