Test set-up for the silicon battery: the battery itself is only the size of a button cell and is located in the hollow cylinder in the middle of the acrylic glass casing. The thin channels that pass through the housing control the supply and outlet of the electrolyte fluid. Credit: Forschungszentrum Jülich
Silicon-air batteries are viewed as a promising and cost-effective alternative to current energy storage technology. However, they have thus far only achieved relatively short running times. Jülich researchers have now discovered why.
In theory, silicon-air batteries have a much higher energy density and are also smaller and lighter than current lithium-ion batteries. They are also environmentally friendly and insensitive to external influences. Their most important advantage, however, is their material. Silicon is the second most abundant element in the Earth's crust after oxygen: it is cheap and its reserves are practically inexhaustible.
However, the silicon-air battery does still have a few crucial blemishes: for example, the flow of current stops after a relatively short period of time. Only assumptions have been made thus far as to why this is the case: does a protective layer form spontaneously on the silicon anode? Is the electrolyte at all suitable? Is there a problem with the air electrode? Attempts to rectify this problem by improving the components have proven to be less than successful. The best result was achieved through the use of a special, high-quality electrolyte based on an ionic liquid. This helped increase the battery's running time to several hundred hours, but contradicted the fundamental idea of the battery: to provide a cost-effective alternative to lithium-ion batteries.
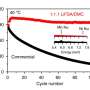
Scientists at Jülich's Institute of Energy and Climate Research (IEK) suspect another cause for the short running time: the consumption of the electrolyte. As part of the AlSiBat project funded by Germany's Federal Ministry of Education and Research, the researchers developed a pump system in which the electrolyte fluid – potassium hydroxide dissolved in water – was refilled from time to time. "If the silicon anode remains in contact with the electrolyte, the battery will continue running," explains Hermann Tempel from the IEK's Fundamental Electrochemistry. The battery is thus able to achieve a running time of over 1,100 hours, or almost 46 days, he adds. "Until the silicon is fully used up. The battery can subsequently be recharged by exchanging the anode, in other words mechanically."
The scientists are now looking for a way to keep the battery running without having to refill the electrolyte. "We need to stop the battery from self-discharging," explains Hermann Tempel, noting how this leads to the electrolyte fluid being used up. Additives in the electrolyte could help here, he says. "The battery is not yet perfect, but we now know what we have to work on."
Provided by Forschungszentrum Juelich