Researchers dispute reported breakthrough in lithium-battery technology

Recent research published in the prestigious journal Science described a breakthrough in lithium-battery technology—the super-efficient source of power for everything from smartphones and laptops to electric cars. The researchers, from Cambridge University, claimed success at engineering a "lithium-air," or Li-air, battery, which uses oxygen from the atmosphere as one of its components. It holds the promise of being lighter, longer-lasting, and cheaper than lithium ion, or Li-ion, batteries, but moving the battery from discovery to practical implementation has been a challenge.
However, a number of battery experts, including Northeastern's K.M. Abraham, an inventor of Li-air technology, doubt the team's claims, and penned dissents explaining why in Science last week. We asked Abraham, research professor at Northeastern's Center for Renewable Energy Technologies, to explain the difference between these battery technologies and provide insight into the controversy.
In 1996, you reported on your development of a "novel rechargeable" lithium-air battery in a paper that appeared in the Journal of the Electrochemical Society. How does the lithium-air battery work compared to current lithium-ion batteries?
A Li-ion, battery has three primary parts: Two electrodes—an anode made of graphite and a cathode made of lithium cobalt oxide or a similar metal oxide—and a very thin, but porous, polyethylene separator that keeps the two apart. All of these materials are stored inside the battery. The electric current flows between the anode and the cathode through an external circuit via a liquid called the electrolyte.
A Li-air, battery also has an anode and a cathode. Here, the anode is made of lithium metal but the cathode is oxygen that is accessed from the atmosphere. As with the Li-ion battery, the electric current flows between the anode and the cathode via the liquid electrolyte. But because the cathode is not stored inside the battery, and because oxygen weighs much less than a metal oxide cathode, a Li-air battery is extremely light.
Currently Li-air batteries remain in the early stages of development rather than in practical use. If a Li-air battery were to become practical, it would provide three to four times the energy of a Li-ion battery. In the best-case scenario, a Li-ion battery stores about 240 watt hours per kilogram of battery weight. The Li-air, on the other hand, would provide more than 1,000 watt hours per kilogram of battery weight.
What would lithium-air batteries enable us to do that lithium-ion ones can't regarding technologies such as electric cars?
Oxygen is environmentally friendly: It is nontoxic, plentiful, and as I mentioned earlier, much lighter than other materials, reducing drag. Any device—a smartphone, iPad, electric vehicle—could therefore run much longer than with a Li-ion battery. For example, an electric car powered by a Li-ion battery can travel about 200 to 250 miles on a single charge, whereas one powered by a Li-air battery could readily go from 400 to 500 miles on a single charge.
The military is interested in Li-air batteries for strategic reasons. In addition to being long lasting, they are easy to carry, so, for instance, a soldier in a remote area could simply open a package and expose the batteries to the air. Commercial applications include storing the electricity generated by wind turbines and solar panels.
A research team at Cambridge University described a breakthrough in lithium-air technology that you and researchers at six other universities or national laboratories contested in two dissents in Science last week. What advances are claimed in the paper, and what were the issues that the dissenters had with them?
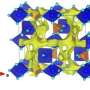
The Cambridge researchers modified the Li-air cell chemistry by adding lithium iodide and a small amount of water into the battery electrolyte as well as using a graphene oxide electrode instead of a carbon one. They claimed an improved charge/discharge cycle life for their modified Li-air cell, meaning their battery could be charged many more times the current Li-air battery, extending its lifespan. However, based on the thermodynamic considerations we addressed in our Science commentary, the data they presented in their earlier Science paper could not be attributed to a Li-air battery but rather to a lithium-iodine battery, another high-energy rechargeable battery but without the potential of Li-air technology. Bottom line, in the Cambridge paper we found the claims about the chemistry not sound.
More information: T. Liu et al. Cycling Li-O2 batteries via LiOH formation and decomposition, Science (2015). DOI: 10.1126/science.aac7730
Journal information: Science
Provided by Northeastern University