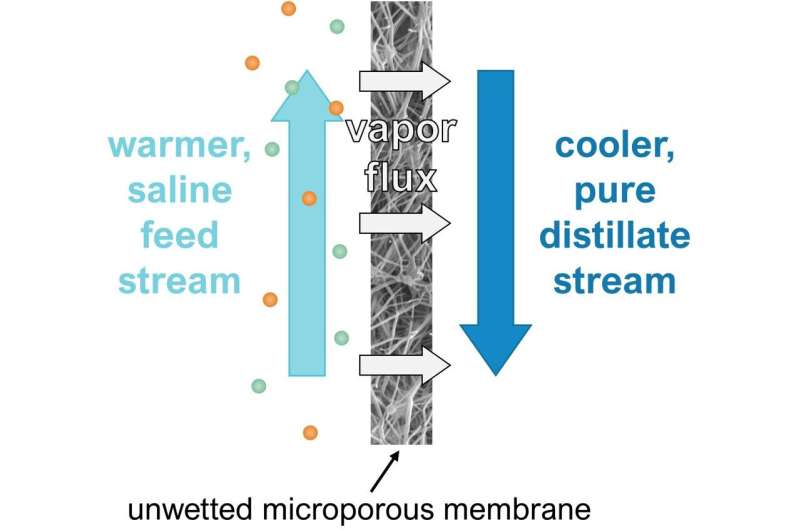
The warm high salinity water stream is on one side of the membrane and a cool pure water stream is on the other. the warmed water evaporates, leaving the salt behind and forming a vapor, which transforms to a distilled liquid form when meeting the cool water stream on the other side of the membrane. Credit: Allyson McGaughey.
Growing up in Seattle, Allyson McGaughey, USC Viterbi School of Engineering Ph.D. '21, was never faced with the daily reality of drought. In the increasingly hot and dry desert of Los Angeles, however, the scarcity of water was laid bare—increasing the urgency of finding alternate water solutions.
In research published in the Journal of Membrane Science, McGaughey, in coordination with Amy Childress, USC Viterbi Gabilan Distinguished Professor, revealed new insights on how best to design water purification processes—for example, wastewater treatment at a water treatment facility—using membrane distillation (MD). MD is a process which separates salt from water using a thin, dry, porous membrane. Moderate temperature differences drive water to pass from one side to another.
To understand this better, think of a spaghetti strainer, but with much, much smaller holes. A water stream poured through the strainer will be "cleaned" of certain materials in the water that are too big to pass through the strainer's holes (like a membrane's pores), leaving a "clean" stream on the other side of the strainer. Still, anything smaller than those holes—like the dissolved salt in our pasta water—can still get through. To purify even further, what if we could collect just the steam, or pure water vapor? Now, imagine a strainer that only allows steam, not liquid water, to pass through the holes. Then, even dissolved salts can't get through. By using a very hydrophobic (water-fearing) membrane that does just that, MD can be used to extract pure, desalinated water from contaminated streams.
The success of membrane distillation, the researchers said, relies largely on membrane designs that can reduce or eliminate moisture accumulating in the membrane. If a membrane does become wet, the researchers said, it can lose its efficacy, compromising the quality of the treated water. To this end, McGaughey, now a post-doctoral fellow at Princeton University, studied how best to design membranes so they do not become excessively wet and successfully treat water, eliminating salt and contaminants and creating a high quality, or pure, outflow.
Among their key findings, McGaughey said, are that reducing membrane pore size or increasing thickness of the membrane itself can increase water resistance and delay or prevent contamination of the purified water stream.
Membranes are typically made of a hydrophobic, or water resistant, synthetic material with pores 0.1 to 0.5 micrometers small. McGaughey said that while other processes are typically more energy efficient than membrane distillation—for example, a process called reverse osmosis—in the case of saltier water streams, these more typical processes require a formidable amount of pressure to force the water molecules through the membrane. Thus, making them less practical for treating very salty streams.
By contrast, membrane distillation allows for saltier water to be purified more efficiently than with reverse osmosis and allows scientists to purify saltier wastewater that is usually disposed of because it can't be efficiently cleaned by traditional water treatment processes.
The problem, McGaughey said, is that the membranes that filter the wastewater can get excessively wet. "In reverse osmosis we use dense membranes that are nonporous so only water molecules get through, but in membrane distillation there are holes in the membranes that can allow for contamination if they get wet," she said.
Optimizing membrane distillation to increase water resistance of membranes
Desalination is inherently a costly and energy-intensive process due to the chemical properties of salt and water. Salt easily dissolves in water, making bonds that are very difficult to break, the researchers said.
"If we had a choice, we wouldn't be desalinating at all," McGaughey said, "but increasingly we need that water."
With membrane distillation, McGaughey said a warmed salty stream is placed on one side of a dry membrane and a cool, pure water stream is on the other. The difference in temperature between the two streams is the driving force that moves water from one side to another. In order to separate pure water from salt and other contaminants, the water molecules in the salty stream shift from a liquid into a vapor gas due to the heat.
Inside the dry membrane pores, there is a small air gap that allows for vapor collection, which occurs when the saltwater is warmed and evaporates, passing through the membrane while leaving the salt behind. Because the air gap is small, not a lot of heat is needed to change the saltwater into vapor, meaning you can use solar energy to warm the salty liquid. The vapor represents the purified water or distillate, which on the other side of the membrane, is cooled —by the cold water—and returns to a liquid form.
The membrane's resistance to liquid water, or wetting resistance, is key to ensuring that the distillate stream is actually purified versus contaminated. When the membrane gets wet, liquid water mixes from the wastewater, or saline stream, into the purified water stream, creating a lower quality output—perhaps even a water output that would not meet potability standards.
Trying to figure out how a membrane loses its wetting resistance on a fundamental level and how this can be prevented through hydrophobicity of the membrane material and pore size is key, the researchers said.
"We have membranes that work now, but when you go up to extremely high salinities, and you get salt precipitation on the membrane surface, that's still a big challenge," McGaughey said.
Emerging challenges in water supply
"Managing high salinity waste streams is a major challenge—for example, industrial waste streams," McGaughey said.
"It [membrane distillation] is never going to be more energy efficient than reverse osmosis, but it can use solar thermal power or low-grade 'waste' heat, meaning it can rely on green energy. This means less carbon emissions than the electricity we use to drive reverse osmosis and it can reach higher salinity streams as well," she said.
Instead of any single process being a standalone solution, McGaughey said membrane distillation could be a complement to reverse osmosis, for instance something you can use downstream (further in the water treatment process), following a reverse osmosis treatment.
"Membrane distillation could be used on the rejected saltwater stream that comes out of reverse osmosis to maximize use of the available water," she said.
McGaughey also said membrane distillation could have applications in rural and non-electrified regions as well.
More information: Allyson L. McGaughey et al, Wetting indicators, modes, and trade-offs in membrane distillation, Journal of Membrane Science (2021). DOI: 10.1016/j.memsci.2021.119947
Provided by University of Southern California