This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Spectroscopic technique that singles out water molecules lying on the surface reveals how they relax after being excited
A more complete picture of how excited water molecules at an interface with air lose their energy has been uncovered by RIKEN scientists in a study published in the journal Nature Communications. This finding will be valuable for better understanding processes that occur at water surfaces.
Water is an anomaly in many ways. For example, its freezing and boiling points are much higher than might be expected, and it is less dense as a solid (ice) than as a liquid.
Almost all of water's unusual properties stem from weak bonds that are continually forming and breaking between neighboring water molecules. Known as hydrogen bonds, these bonds arise because oxygen attracts electrons more than hydrogen. The slightly negative oxygen in one molecule is thus attracted to the slightly positive hydrogens in other molecules.
But a tiny sliver of water molecules--those lying on the surface--experience hydrogen bonds differently than do other water molecules. In their case, the arm sticking up into the air doesn't form hydrogen bonds.
Until now, no one had been able to discover how the arms of these surface molecules relax after being stretched. That's because it is incredibly challenging to isolate the signal from these molecules.
"We have a good knowledge of how water molecules in the body of the liquid behave, but our understanding about water molecules at the interface lags far behind," says Tahei Tahara of the RIKEN Molecular Spectroscopy Laboratory.
Over the past decade, a team led by Tahara has been trying to rectify this situation by developing highly sophisticated spectroscopy techniques to probe the interactions of water molecules at surfaces.
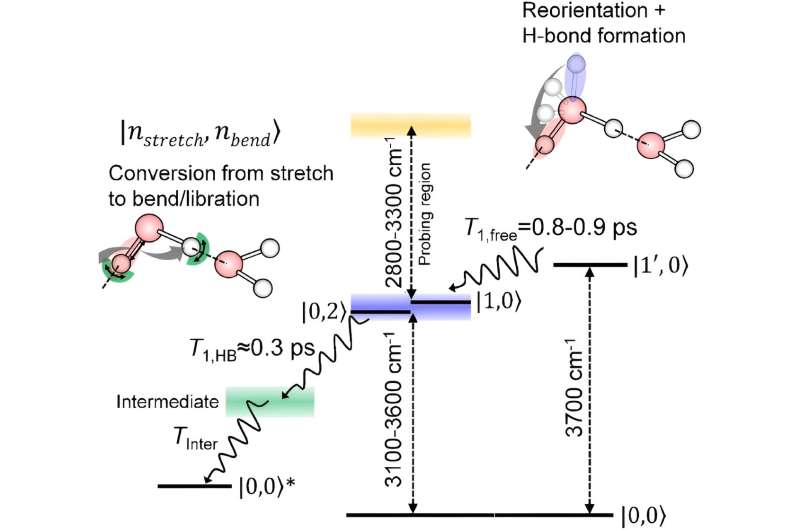
The team has now developed a technique based on infrared spectroscopy that is sensitive enough to detect how the oxygen–hydrogen bonds of surface water molecules relax.
Using the technique, the team found that oxygen–hydrogen bonds sticking up into the air first rotate without losing energy. They then relax in a similar way to molecules in the body of the liquid that form a hydrogen-bond network.
"In this sense, there is no big difference between molecules at the interface and inside the liquid after interaction with neighbors—they both share the same relaxation process," says Tahara. "These findings paint a comprehensive picture of how stretching of oxygen–hydrogen bonds relaxes at the surface of water."
Tahara and his team now intend to use their spectroscopic technique to look at chemical reactions that occur at the interface of water.
More information: Woongmo Sung et al, Unified picture of vibrational relaxation of OH stretch at the air/water interface, Nature Communications (2024). DOI: 10.1038/s41467-024-45388-8
Journal information: Nature Machine Intelligence , Nature Communications
Provided by RIKEN