This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Novel synthesis of fluorinated molecules with potential in drug research
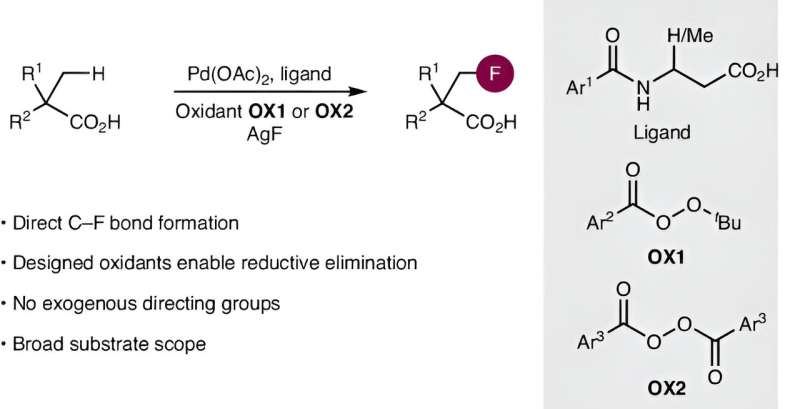
Carboxylic acids are one of the most important substance classes in chemistry and are a component of many drugs such as aspirin and ibuprofen. To tailor the properties of carboxylic acids, fluorine atoms can be introduced into the molecular structure. However, this requires complex, multi-step synthetic routes.
In the journal Nature Synthesis, an international team of researchers from the Otto Diels Institute of Organic Chemistry at Kiel University (CAU) has now published an article describing a method for the direct introduction of fluorine atoms into aliphatic carboxylic acids that significantly simplifies and accelerates the process.
Innovative combined approach addresses fundamental challenge
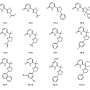
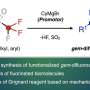
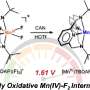
The new synthetic method relies on two fundamentally challenging chemical processes. First, a typically unreactive carbon–hydrogen bond must be activated, in this case by cleavage with a palladium catalyst ("C–H activation").
In recent years, several international research teams have laid the foundations for suitable catalysts, including the group led by Manuel van Gemmeren, Professor for Organic Chemistry at CAU. Based on this preliminary work, they have developed new, particularly efficient catalysts in their current publication.

The second key challenge of this work was to form a carbon–fluorine bond. Established strategies proved to be unsuitable for aliphatic carboxylic acids. Van Gemmeren's team therefore developed an innovative new approach. "In addition to optimizing the catalyst structure, we designed a reagent, a so-called oxidizing agent, that influences the reaction in the crucial step and thus enables the selective formation of the carbon-fluorine bond," explains the first author Sourjya Mal.
Further experiments confirmed that an unusual reaction pathway is indeed operative using these reagents. "In this study, we were able to show that the combined design of catalyst and oxidant enables reactions that would not be feasible with a single approach. This finding is certainly transferable to other highly attractive synthetic methods and could therefore prove to be groundbreaking," says van Gemmeren.
Synthetic method with extraordinary potential for pharmaceutical research
With the reported method, fluorine atoms can be introduced directly into complex carboxylic acid molecules without a tedious and time-consuming synthesis.
"Due to the importance of both carboxylic acids and fluorinated molecules in pharmaceutical research, I see extraordinary application potential for our method," says Professor von Gemmeren. "I am very proud of what my colleagues, especially the first author of the study, Sourjya Mal, have achieved here."
More information: Sourjya Mal et al, Pd-catalysed direct β-C(sp3)–H fluorination of aliphatic carboxylic acids, Nature Synthesis (2024). DOI: 10.1038/s44160-024-00578-6
Journal information: Nature Synthesis
Provided by Kiel University