December 2, 2021 report
Using Raman spectroscopy and computational techniques to study interfacial water on Pd single-crystal surfaces
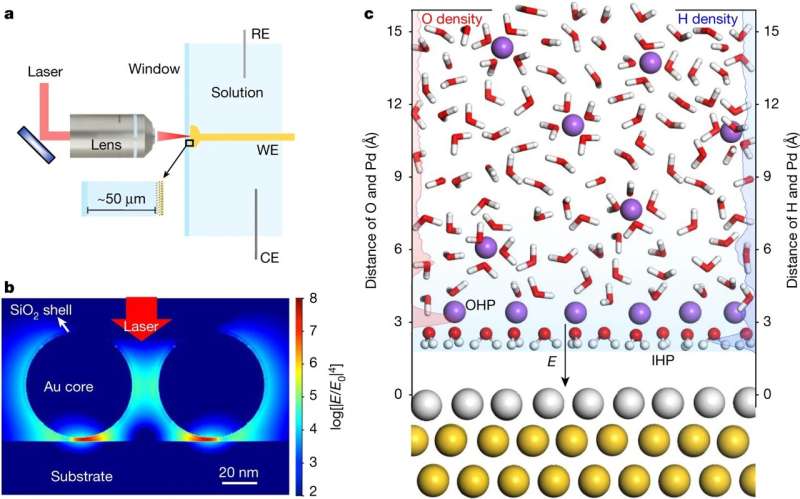
A team of researchers affiliated with a number of institutions in China and one in the U.K. has used Raman spectroscopy and computational techniques to study the interfacial water on Pd single-crystal surfaces. In their paper published in the journal Nature, the group describes their study of the dynamics and structure of water at the solid-liquid interface and what they learned from it. Matthias Waegele, with Boston College has published a News and Views piece in the same journal issue discussing the recent history of using spectroscopic techniques to stretch the bonds between hydrogen and oxygen and giving an overview of the work done by the group working on this new effort.
As the researchers note, better understanding the dynamics and structure of water at the solid-liquid interface has become an important component of research in a wide variety of areas, including catalysis. In this new effort, they have expanded on the use of spectroscopic water analysis techniques by using a flat metal electrode (palladium atoms on a gold substrate) to serve as a catalyst. They then added relevant computational techniques to manipulate the spectroscopic data. This allowed for disassociating water in a sample into molecular hydroxide and hydrogen. In so doing they found their technique helped them to probe more deeply into interfacial water samples.
In using their technique, the researchers found that a network of water molecules worked to connect intermolecular hydrogen bonds that surrounded the electrode under low sodium ion concentrations. They also found that at higher concentrations, hydrated sodium ions were attracted to the electrode which resulted in bringing the hydrogen and palladium atoms closer together. And that led to speeding up the transfer of electrons between the water molecules and the electrode which led to the disassociation of the water into hydroxide ions and hydrogen atoms. That further led to the formation of hydrogen molecules. The researchers note that this finding suggests their technique could be used as a new form of electrocatalysis—a means of accelerating chemical reactions that result in converting energy from a renewable power source into a green fuel—in this case hydrogen. They also suggest that their technique could be used to speed up the reaction rates of electrocatalytic reactions in general.
More information: Yao-Hui Wang et al, In situ Raman spectroscopy reveals the structure and dissociation of interfacial water, Nature (2021). DOI: 10.1038/s41586-021-04068-z
Matthias M. Waegele, Choreographing water molecules to speed up hydrogen production, Nature (2021). DOI: 10.1038/d41586-021-03511-5
Journal information: Nature
© 2021 Science X Network