Team ensures stability of desalination process with magnesium

A Korean research team found a method to inhibit the fouling of membranes, which are used in the desalination process that removes salt and dissolved substances from seawater to obtain drinking, domestic, and industrial water.
The Korea Institute of Science and Technology (KIST) announced that a research team led by Dr. Seongpil Jeong and Dr. Seockheon Lee at KIST's Water Cycle Research Center developed a membrane distillation pretreatment process that adds magnesium to inhibit the fouling and wetting of membranes during desalination.
The membrane distillation process is a desalination technology used to produce fresh water in which seawater is heated to generate a vapor, which is then passed through a hydrophobic membrane before condensing into fresh water. The phenomena of fouling and wetting can often occur during the membrane distillation process. If fouling occurs, it can cause the production of fresh water to take much longer or shorten the lifespan of the membrane used in the distillation process, thereby increasing the costs associated with freshwater production.
The KIST research team monitored the membrane distillation process and found that the formation of calcium carbonate (CaCO3) and calcium sulfate (CaSO4) crystals on the membrane surface was the main cause of fouling. They also found that the formation of CaCO3 crystals occurred at the beginning of the membrane distillation process, causing partial membrane wetting, while the formation of CaSO4 crystals caused a complete membrane wetting, halting membrane operation.
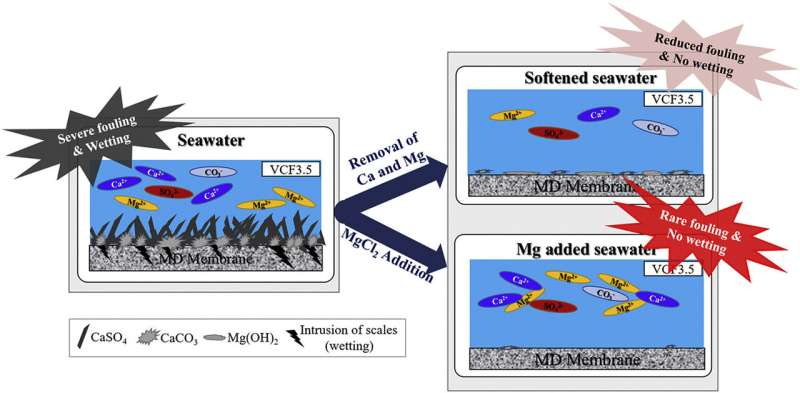
Anti-scalants are often applied to prevent fouling caused by calcium-based crystal growth in the desalination process. There have been some reports in the industry about a chemical softening technology that can be applied as a pretreatment process for membrane distillation. However, it has also been reported that organic anti-scalants can reduce the surface tension of the feed solution and cause wetting. The application of chemical softeners, however, requires additional large-scale sedimentation and filtering processes to remove the crystals formed during the softening process.
KIST's research team, led by Dr. Seongpil Jeong, is the first research team to develop a pretreatment process that adds magnesium to seawater. The team found that magnesium inhibits the formation of CaCO3 and CaSO4 and effectively prevents membrane fouling since the magnesium readily combines with carbonate and sulfate ions in the seawater. The team also found that the added magnesium chloride (MgCl2) increased the stability of the hydrophobic membrane, thereby increasing wetting resistance.
"Increasing the stability of the hydrophobic membrane is expected to help improve desalination efficiency and lengthen the lifespan of the membrane," said Hye-won Kim first author of the Water Research paper. "The mineral-based, environmentally-friendly pretreatment is applicable not only to the membrane distillation process but also to various other desalination processes."
More information: Hye-Won Kim et al, Retardation of wetting for membrane distillation by adjusting major components of seawater, Water Research (2020). DOI: 10.1016/j.watres.2020.115677
Provided by National Research Council of Science & Technology