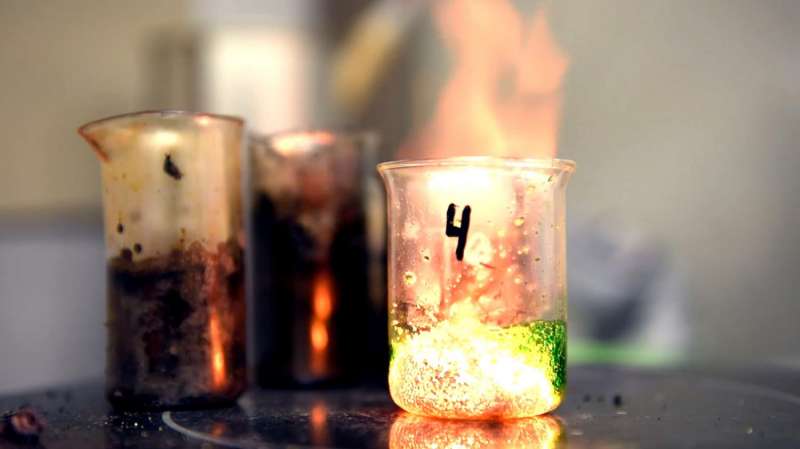A group of NUST MISIS scientists led by Professor Alexander Mukasyan has produced a unique accelerator by developing self-propagating high-temperature synthesis (SHS). The accelerator doesn't degrade or become polluted while working, and therefore operates 10 times longer than ordinary accelerators. It has been running continuously for several years, and is jokingly called "eternal." Accelerators are used to obtain nanomaterials, as well as for the post-combustion process in vehicles. Additionally, they reduce harmful emissions.
The creation of nanoscale materials with desired properties is accompanied by a number of difficulties. The majority of methods don't allow workers to get the final material with the desired nano-sizes (for example, a size less than 10 nm is important from the magnetic characteristics point of view) and/or high specific surface (which influences the catalytic activity). The creation of the majority of nanomaterials requires special, complicated equipment and high-energy consumption.
Self-propagating high-temperature synthesis (SHS) in solutions or "solution combustion" is an alternative method of nanomaterial synthesis. Key to the system is the self-sustaining exothermic reaction (combustion) of components' interaction based on systems containing oxidants (metal nitrate) and reductive agents (water-soluble linear and cyclic organic amines, acids, and amino acids). The chemical reaction is intensively distributed in the solution, and as it fades away, the final products are formed; then the single process of combustion and the receipt of materials occurs. Initial reagents are mixed in solutions at the molecular level, and the large amount of gas emission during the interaction with reagents in a combustion wave facilitates the formation of nanopowders with desired characteristics.
Scientists of NUST MISIS's Center of Functional Nano-Ceramics have achieved impressive results by studying materials' synthesis combustion solutions using a methodology of solution physics and chemistry. By placing a mixture of nickel nitrate and glycine in a highly porous medium and running the reaction, they obtained a new type of super-stable accelerator which doesn't degrade and doesn't become polluted during the working process. A review of the research in this field has been published by Chemical Reviews.
"Our research will allow us to bring light to mechanisms underlined by a basis of synthesis of nanomaterials by SHS solutions. Behind the simple and beautiful process are hidden complicated mechanisms, and though the nature of it all is very difficult to understand, researchers will be able to obtain new nanomaterials with amazing properties," noted Professor Alexander Rogachev, vice-director of the NUST MISIS Center of Functional Nano-Ceramics.
This opens a wide range of opportunities for the development of modern energy. The final materials will be used in fuel and solar cells, capacitors and batteries of a new generation, and even in thermoelectric materials (used in thermoelectric generators to convert heat to electricity). They are in demand for hydrogen energetics—for example, for hydrocarbon conversion into methane or for the production of pure hydrogen from ethanol—and can be used as phosphors — substances that are able to convert absorbed energy into light emission.
Journal information: Chemical Reviews