In nearly every organism's genome, scattered between genes that encode proteins, long regulatory regions stretch across expanses of DNA. Understanding what role these so-called enhancer regions play in controlling the activation of genes has been a slow process for scientists—methods to study enhancers have required altering their genetic sequence or moving them from their normal spot in the genome. Now, Howard Hughes Medical Institute scientists have developed a new way to study enhancers, by using proteins called transcription activator-like effectors, or TALEs, to bind the regions of DNA.
"We've had a pretty vague understanding of how enhancers work," says HHMI Janelia Farm Research Campus group leader David L. Stern, who studies the genetic mechanisms that underlie organisms' diversity and spearheaded the new research. "Now we have the ability to target many different factors to these regions and start asking more detailed questions about their biology."
Previous studies have established that when transcription factor proteins bind enhancer regions, patterns of expression in genes—either close by or far away—change. The binding of activators or repressors to the enhancers controls whether or not a gene in any given cell is expressed. This kind of genetic regulation allows for the specialization of different cell types in the body—letting neurons and skin cells each turn on the genes they need to carry on their unique functions —even though both contain the same genetic information. Many disease-causing mutations and genetic changes contributing to evolution have been traced to enhancer regions of DNA, demonstrating that the changes in these regulatory regions can have significant impact on an organism. But scientists have an incomplete understanding of how enhancer sequence encodes function, how different transcription factors interact with each other while binding enhancers, and how this protein binding causes changes to gene expression.
In an attempt to better understand enhancers, Stern and Justin Crocker, a researcher in his lab, turned to TALE proteins. TALEs were originally isolated from bacteria that infect plant cells and alter plants' gene expression. In 2009, researchers in Germany and the U.S. discovered the code that mediates what gene sequence a TALE binds to, allowing the development of TALEs that bind to any sequence of interest.
"We can just go in and reorganize particular repeats in the TALEs so that they bind to whatever target sequence we want," says Crocker.
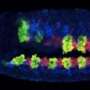
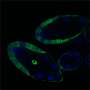
So Stern and Crocker designed TALEs that recognized the sequence of the enhancer regions for the gene even-skipped (eve), which has been well-characterized in the fruit fly, Drosophila melanogaster. Then, they attached the repressor domain of the Hairy protein to the TALE. Adding this TALE to a developing fly embryo, they hoped, would localize the Hairy repressor to theeve enhancer.
"The Hairy repressor and the eve locus are classic systems, so we chose them to ensure that using TALEs, we could replicate what we already knew about the eve enhancers," says Stern. "In truth, we weren't sure what would happen—would a single repressor protein be able to shut down a single enhancer?" says Justin.
The TALE-repressors worked: when the engineered TALEs were added to embryos, the levels of eve expression plummeted, becoming almost undetectable. When they added a gene activator, instead of the Hairy repressor, to the TALE, eve expression skyrocketed. It's the first example of direct binding to an unaltered enhancer in a living organism to alter gene expression. In both cases, no aspect of development unrelated to the targeted enhancer of eve was apparently affected, suggesting that the binding was extremely specific to the enhancer it was designed for.
This technique can now be used to test enhancer regions that haven't been characterized before and to further probe enhancer function. Scientists can use engineered TALEs to bind any DNA region, with either a repressor like Hairy or an activator, and then study the resulting changes in an organism's gene expression.
"Not only does this let us understand enhancers in their native context," says Crocker, "but it lets us take the next step forward to controlling them."
Crocker and Stern are now fine-tuning the TALEs so that they have even more control over DNA binding and regulation of enhancer activity, and they are planning to use the technique to study other enhancers.
Provided by Howard Hughes Medical Institute