Credit: Unsplash/CC0 Public Domain
A research group led by Profs. Bao Xinhe, Wang Guoxiong and Gao Dunfeng from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS) has revealed the mechanism of coverage-driven selectivity switch from ethylene to acetate in high-rate CO2/CO electrolysis.
The study was published in Nature Nanotechnology on Jan. 12.
Electrolysis of CO2 to valuable chemicals and fuels with power supply derived from renewable energy has emerged as a promising carbon capture, utilization, and storage technology.
To push the CO2 electrolysis process towards practical application, high selectivity of multicarbon (C2+) products at industrial current densities is highly desirable. Moreover, tuning the microenvironments near catalyst surfaces has also been demonstrated to be effective in facilitating C–C coupling and improving C2+ production.
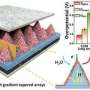
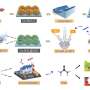
In this study, the researchers tuned the catalyst microenvironment using mixed CO/CO2 feeds that were typical composition of waste gases from steel plants and incomplete industrial combustion of fossil fuels. Specifically, they investigated CO/CO2 co-electrolysis over a nanoporous CuO nanosheet catalyst in an alkaline membrane electrode assembly (MEA) electrolyzer at high current densities.
"With increasing CO pressure in the feed, the major product gradually shifted from ethylene to acetate and the current density remarkably increased, up to 3.0 A cm-2 under 0.6 MPa pure CO feed," said Prof. Wang.
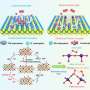
"The selectivity switch was induced by *CO coverage and local pH. Ethylene was preferentially generated at low *CO coverage, whereas acetate formation was favorable at high *CO coverage and high local pH," said Prof. Gao.
The researchers further improved electrolysis performance by optimizing electrolysis conditions. The Faradaic efficiency and partial current density of C2+ products reached 90.0% and 3.1 A cm-2, corresponding to a carbon selectivity of 100.0% and yield of 75.0%, outperforming thermocatalytic CO hydrogenation.
The scale-up of the CO electrolysis process was demonstrated using an electrolyzer stack composed of four 100 cm2 MEAs, with the highest ethylene formation rate of 457.5 mL min-1 at 150 A and acetate formation rate of 2.97 g min-1 at 250 A.
"Our work highlights the promise of tuning catalyst microenvironments for selective production of single C2+ products such as acetate and ethylene, and presents an effective scale-up demonstration of high-rate CO2/CO electrolysis towards practical application," said Prof. Wang.
More information: Guoxiong Wang, Coverage-driven selectivity switch from ethylene to acetate in high-rate CO2/CO electrolysis, Nature Nanotechnology (2023). DOI: 10.1038/s41565-022-01286-y. www.nature.com/articles/s41565-022-01286-y
Journal information: Nature Nanotechnology
Provided by Chinese Academy of Sciences