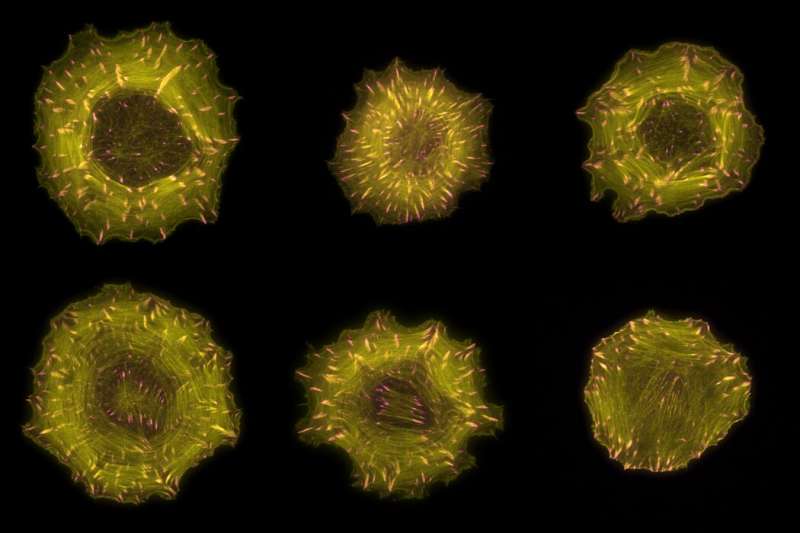
Microscopic images of cells in which filaments of the actin cytoskeleton are labelled in yellow and focal adhesions are labelled in pink. Credit: Markus Müller, Rocks Lab, MDC
The cytoskeleton is a permanent construction site consisting of protein filaments that are continually lengthening and shortening in a dynamic process. Through these remodeling processes, the cell can change its shape and even move to a new location. In this way, it guides fundamental processes, such as cell division and differentiation, and processes at a higher level in the organism, such as embryonic development and wound healing. If something goes wrong at the cytoskeletal construction site—e.g., if protein filaments undergo remodeling at the wrong place or time—it could lead to diseases. Such an error in spatio-temporal control is also the reason why metastatic cancer cells migrate in the body.
Researchers at the Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC) and other institutes, such as the European Bioinformatics Institute (EMBL-EBI) in Hinxton, UK, have investigated how a family of 145 proteins causes these remodeling events to occur at the right place and time. Up to this point, scientists had only examined these regulatory proteins in individual studies, and only a few proteins had been characterized.
"In order to understand complex processes, including cell shape changes, we need to know how the regulatory proteins work together collectively. Until now, we lacked a bird's eye perspective, so to speak," says Dr. Oliver Rocks, head of the research group and senior responsible author of a new study in the journal Nature Cell Biology. His group, with lead authors Dr. Paul M. Müller and Dr. Juliane Rademacher, together with the group led by Dr. Evangelia Petsalaki at EMBL-EBI and an international research team, has now systematically characterized all these regulatory proteins. The team was able to show that there are different signaling zones within the cell that coordinate the cytoskeleton in space and time and also how these zones are created and maintained.
A new perspective, thanks to a comprehensive database
At the cytoskeletal construction site, the Rho GTPase proteins set the tone. When these molecular switches are activated, they send commands to the machinery on site. There are 145 regulatory proteins that control these molecular switches: RhoGEF proteins turn them on, RhoGAP proteins turn them off. Now Rocks and his team have systematically investigated all these regulators for the first time and created a kind of library. Researchers from all over the world can access this library to see which molecular switches individual proteins control, where in the cell this occurs and which binding partners it has.
The comprehensive information contained in the protein library allows proteins to be analyzed at the systems level for the first time—i.e., from a bird's-eye perspective. This has revealed new collective properties of the regulators that were previously imperceptible. In this way, researchers discovered a new mechanism that explains how cell migration is controlled.
Focal adhesions control the balance of two opposing processes
Scientists were aware that two opposing processes controlled by the cytoskeleton—cell protrusion and cell contraction—need to occur in separate places in the cell to allow it to move. In one construction site at the front of the cell, the molecular switches give the command for cell protrusion in the migration direction. Further behind, toward the cell interior, they trigger a contraction of the cytoskeleton. The central question investigated by the MDC team was how Rho GTPases coordinate these two spatially separated processes.
The spatial organization of the two opposing processes is made possible through focal adhesions, explains Rocks. These are protein accumulations located directly below the cell membrane that anchor the cell to its environment. Focal adhesions occur close to the front of the cell, mature into more stable structures and eventually dissolve again. As the cell travels through focal adhesions during migration, these move from the front to the middle of the cell.
"The cell exploits the fact that focal adhesions change their location," Rocks says. Initially, his team discovered a surprising number of regulatory proteins on these structures. But the real surprise, he says, "was that we found a specific subgroup of regulators located almost exclusively on newly formed focal adhesions at the cell's edge and another separate subgroup on mature structures toward the middle of the cell." These subgroups control the opposing cytoskeletal processes mentioned above, creating the spatially separated signal zones. The researchers were also able to show that both processes are linked by mechanical forces in the cell, which can help maintain a balance between the number of newly formed focal adhesions and the number of mature ones.
Rocks next plans to investigate how precisely the collective Rho GTPase regulators on the focal adhesions communicate with the cytoskeletal machinery, whether the principle of spatial separation of these proteins also plays a role on other cell structures and how a defective regulatory function can ultimately lead to diseases.
Rocks confirms that gaining this systemic view of cell shape regulation has taken a great deal of effort. "However, it was absolutely essential to revitalize this research field and open up new conceptual approaches for further study," the researcher says. The database and protein library are now available to all scientists worldwide.
More information: Paul M. Müller et al, Systems analysis of RhoGEF and RhoGAP regulatory proteins reveals spatially organized RAC1 signalling from integrin adhesions, Nature Cell Biology (2020). DOI: 10.1038/s41556-020-0488-x
Journal information: Nature Cell Biology
Provided by Max Delbrück Center for Molecular Medicine