Novel microscopy technique developed to analyze cellular focal adhesion dynamics
Focal adhesions are large specialized proteins that are located in the area where a cell membrane meets the extracellular matrix (ECM), a collection of molecules surrounding the cells that provide support and regulate micromechanical signals to the cells. Examining focal adhesions is one of the key elements to understanding how a cell proliferates, differentiates, and migrates—which can help in the treatment of diseases like cancer.
Researchers at the Beckman Institute for Advanced Science and Technology and the Micro and Nanotechnology Laboratory at the University of Illinois have developed a new form of microscopy that allows them to observe the formation and evolution of cell membrane focal adhesions. Their paper, "Quantitative Analysis of Focal Adhesion Dynamics Using Photonic Resonator Outcoupler Microscopy (PROM)" details how the new live cell imaging technique can observe the formation and evolution of cell membrane focal adhesions. It was recently published in Light: Science & Applications.
"This is a new kind of biophysics method used to measure the peak intensity shift (PIS) of the spectra reflected from the biomaterials on a photonic crystal surface," said Yue Zhuo, a Beckman Institute Postdoctoral Fellow and first author on the paper. "The PIS indicates the variation of cluster size in the focal adhesion area of the cell while it's alive."
Previous methods involve labeling the cells with fluorescent dyes or tags, which not only can change the physical and chemical makeup of the cell, but also can prove cumbersome for researchers. "Typically people look at focal adhesions with fluorescent tags or proteins," Zhuo said. "But fluorescent imaging is an invasive imaging method that may change the conformations or block the binding sites of the proteins in the focal adhesion area."
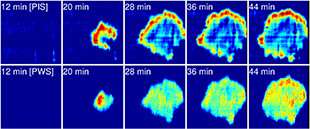
Fluorescence microscopy is severely limited by an effect called "photobleaching," in which the fluorophores only maintain their brightness for several seconds. However, many important cell processes occur over the course of minutes, hours, or days. Because PROM does not use fluorophores, and only uses low-intensity illumination, there is not a limit to how long live cells can be measured.
"In the future, we plan to use PROM to study stem cell differentiation, which can occur over the course of several weeks," said Zhuo.
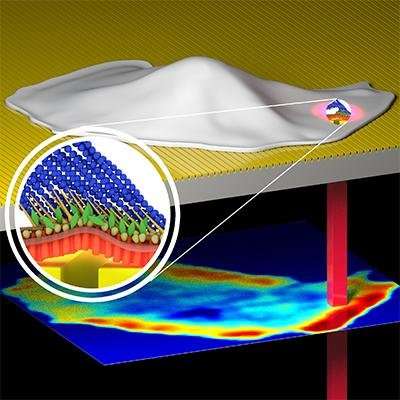
PROM utilizes a photonic crystal biosensor surface to create an evanescent field (a surface-bound electromagnetic field), which selectively illuminates only the ECM-attached cell membrane and associated protein aggregates directly inside the cell membrane. The photonic crystal strictly limits lateral propagation of light while keeping light tightly bound to the biosensor surface, to enable high-resolution imaging of the cell membrane attachment footprint.
"PROM is providing real-time information about dynamic processes that occur specifically on cell membranes that is not available by any other method," said Brian Cunningham, a professor of electrical and computer engineering and bioengineering, and the principle investigator for the PROM project. "Since so many biological processes are mediated through attachment of cells to surfaces, PROM provides a unique view of migration, chemotaxis, chemotoxicity, differentiation, biofilm formation, and division. We see PROM as an exciting new tool for cell biologists that can also be applied towards personalized anticancer drug selection, tissue engineering, and sensor-integrated tumor modeling."
More information: Yue Zhuo et al, Quantitative analysis of focal adhesion dynamics using photonic resonator outcoupler microscopy (PROM), Light: Science & Applications (2018). DOI: 10.1038/s41377-018-0001-5
Journal information: Light: Science & Applications