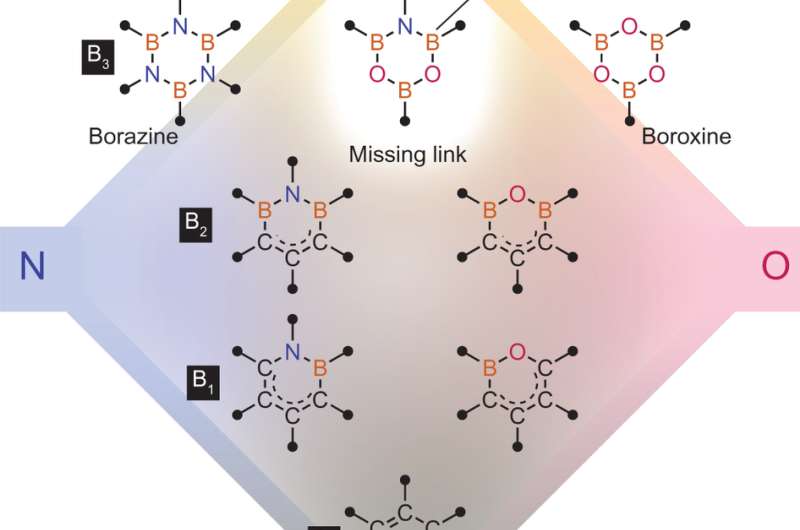
Chemical space for six-membered heterocycles composed of B, C, N and O atoms and the structure of DATBs. Credit: (c) Nature Chemistry (2017). DOI: 10.1038/nchem.2708
(Phys.org)—Polycyclic aromatic hydrocarbons, such as graphene, have several potential uses as its π-conjugated structure lends itself to high stability and conductivity. Researchers are interested in exploring polycyclic aromatic hydrocarbons with boron and nitrogen atoms in an effort to explore the functionality and potential uses for polycyclic aromatic hydrocarbons.
One polyatomic six-member ring that has not been isolated and studied is 1,3-dioxa-5-aza-2,4,6-triborinane, or DATB. DATB has a core B3NO2 ring that evidences some aromatic character. Hidetoshi Noda, Makoto Furutachi, Yasuko Asada, Masakatsu Shibasaki and Naoya Kumagai of the Institute of Microbial Chemistry in Tokyo (Bikaken) isolated and analyzed DATB using an m-terphenyl template. They demonstrated its ability to catalyze the direct coupling of several carboxylic acids with a variety of amines. Their DATB catalyst is thermally stable, structurally planar, and does not undergo hydrolysis. Their work appears in Nature Chemistry.
The m-terphenyl template came out of the necessity to combine the B-N-B fragment to the O-B-O fragment to make the 1,2-dioxa-5-aza-2,4,6-triborinane ring. Using a stepwise synthesis, the first compound they made had the m-terphenyl template attached to the B-N-B portion of the DATB and a phenyl group attached to the boron of the O-B-O portion. The other compound that resulted from their initial syntheses had another m-terphenyl attached to the boron in the O-B-O portion of DATB instead of phenyl.
The presence of DATB was confirmed with multinuclear NMR, mass spectrometry, and single-crystal x-ray diffraction. Notably, the DATB portion is planar and it exhibited offset face-to-face pi stacking. Additionally, it displayed slight aromaticity based on bond length and showed resistance toward hydrolytic cleavage.
The next step was to determine if the DATB derivatives were able to catalyze amide bond formation. Boron has an affinity for amines, making this compound a possible option for catalyzing the direct coupling of amines and carboxylic acids. Noda et al. tested a variety of carboxylic acids because one of the major setbacks with amine and carboxylic acid coupling is that current methods are not generalizable for a variety of carboxylic acids, particularly bulky carboxylic acids.
They tested a range of acids and amines whose coupling reactions were successfully catalyzed using DATB. They found that their DATB derivative successfully catalyzed the amide bond formation for sterically bulky aliphatic and aromatic carboxylic acids. They also found that indole-containing substrates did not require a protecting group. And, their method worked with benzoic acid and primary amines, α-branched amines, cyclic secondary amines. Using higher temperatures, they successfully coupled α-tertiary amines and acyclic secondary amines to benzoic acid. Additionally, they showed that the stereochemistry of α-chiral carboxylic acids and α-chiral amines were maintained.
Their DATB derivative also worked well in the presence of various functional groups showing that the catalyst displayed good selectivity. Additionally, Noda et al. tested the catalyst's ability to make peptide bonds with amino acids and biologically relevant amines by coupling Bz-Val-OH with H-Leu-NH2.
This catalyst is remarkably versatile compared to other catalysts used for amine-carboxylic acid coupling. The authors contend that this is due to the distinct mechanism in which the three Lewis acid sites on the DATB core are used to bind the carboxylic acid and the amine countering the entropy cost in forming the bond.
More information: Hidetoshi Noda et al. Unique physicochemical and catalytic properties dictated by the B3NO2 ring system, Nature Chemistry (2017). DOI: 10.1038/nchem.2708
Abstract
The expansion of molecular diversity beyond what nature can produce is a fundamental objective in chemical sciences. Despite the rich chemistry of boron-containing heterocycles, the 1,3-dioxa-5-aza-2,4,6-triborinane (DATB) ring system, which is characterized by a six-membered B3NO2 core, remains elusive. Here, we report the synthesis of m-terphenyl-templated DATB derivatives, displaying high stability and peculiar Lewis acidity arising from the three suitably arranged boron atoms. We identify a particular utility for DATB in the dehydrative amidation of carboxylic acids and amines, a reaction of high academic and industrial importance. The three boron sites are proposed to engage in substrate assembly, lowering the entropic cost of the transition state, in contrast with the operative mechanism of previously reported catalysts and amide coupling reagents. The distinct mechanistic pathway dictated by the DATB core will advance not only such amidations, but also other reactions driven by multisite activation.
Journal information: Nature Chemistry
© 2017 Phys.org