This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Engineers solve 'catalysis vs corrosion' mystery in electrochemical ozone production
Researchers at the University of Pittsburgh and Drexel University in Philadelphia, along with Brookhaven National Laboratory, are working to solve a multipart mystery to make water disinfection treatments more sustainable.
Scalable electrochemical ozone production (EOP) technologies to disinfect dirty water may someday replace centralized chlorine treatments used today, whether in modern cities or remote villages. However, little is understood about EOP at the molecular level and how technologies that make it possible can be made to be efficient, economical, and sustainable.
Their research, "Interplay between Catalyst Corrosion and Homogeneous Reactive Oxygen Species in Electrochemical Ozone Production," was published recently in the journal ACS Catalysis.
The lead author is Drexel Ph.D. student Rayan Alaufey, with contributing researchers from Drexel, including co-PI Maureen Tang, associate professor of chemical and biological engineering, postdoctoral researcher Andrew Lindsay, Ph.D. student Tana Siboonruang, and Ezra Wood, associate professor of chemistry; co-PI John A. Keith, associate professor of chemical and petroleum engineering, and graduate student Lingyan Zhao from Pitt; and Qin Wu from Brookhaven.
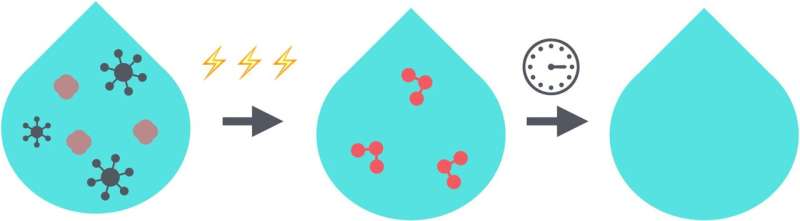
"People have used chlorine to treat drinking water since the 19th century, but today we better understand that chlorine may not always be the best option. EOP for example can generate ozone, a molecule with about the same disinfecting power as chlorine, directly in water.
"Unlike chlorine which stably persists in water, ozone in water naturally decomposes after about 20 minutes, meaning it is less likely to damage people when consuming from water at a tap, when swimming in a pool, or when cleaning wounds in a hospital," explained Keith, who is also R.K. Mellon Faculty Fellow in Energy at Pitt's Swanson School of Engineering.
"EOP for sustainable disinfection would make a lot of sense in some markets, but doing it requires a good enough catalyst, and because nobody has found a good enough EOP catalyst yet, EOP is too expensive and energy-intensive for broader use.
"My colleagues and I thought if we could decode at the atomic level what makes a mediocre EOP catalyst work, maybe we could engineer an even better EOP catalyst."
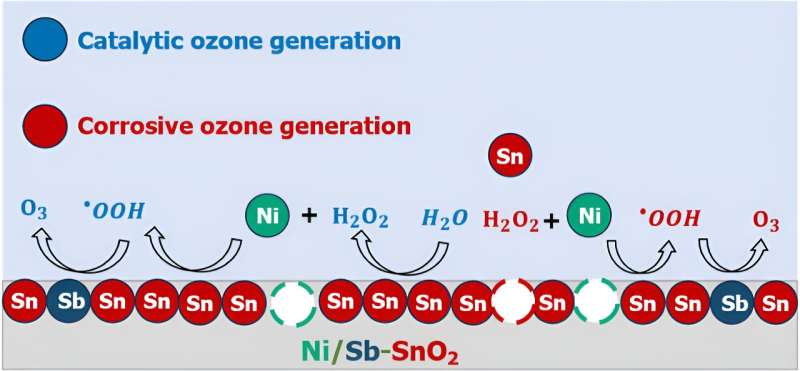
Solving the mystery of how EOP catalysts work is crucial in understanding how to better engineer one of the most promising and least toxic EOP catalysts known to date: nickel- and antimony-doped tin oxide (Ni/Sb–SnO2, or NATO).
Therein, said Keith, lies the conundrum: what is every atom's role in NATO doing to help EOP? Is ozone getting formed catalytically in ways we want it to, or does it form because the catalyst is decomposing, and future work needs to be done to make NATO catalysts more stable?
Surprisingly, the researchers discovered that it is probably a mix of both.
By using experimental electrochemical analyses, mass spectrometry, and computational quantum chemistry modeling, the researchers created an "atomic-scale storyline" to explain how ozone is generated on NATO electrocatalysts.
For the first time, they identified that some of the nickel in NATO is probably leaching out of the electrodes via corrosion, and these nickel atoms, now floating in the solution near the catalyst, can promote chemical reactions that eventually generate ozone.
"If we want to make a better electrocatalyst, we need to understand what parts are working and not working. Factors like metal ion leaching, corrosion, and solution phase reactions can give the appearance that a catalyst is working one way when actually it is working another way."
Keith noted that identifying the prevalence of corrosion and chemical reactions occurring away from the catalyst are important steps to clarify before other researchers can pursue improvements to EOP and other electrocatalytic processes.
In their conclusion, they note, "Identifying or refuting the existence of such fundamental technological constraints will be critical to any future applications of EOP and other advanced electrochemical oxidation processes."
"We know that electrochemical water treatment works on small scales, but the discovery of better catalysts will boost it to a global scale. The next step is finding new atomic combinations in materials that are more resistant to corrosion but also promote economically and sustainably viable EOP," Keith said.
More information: Rayan Alaufey et al, Interplay between Catalyst Corrosion and Homogeneous Reactive Oxygen Species in Electrochemical Ozone Production, ACS Catalysis (2024). DOI: 10.1021/acscatal.4c01317
Journal information: ACS Catalysis
Provided by University of Pittsburgh