January 12, 2017 report
Amino acids formed from the single-electron activation of carbon dioxide
(Phys.org)—Carbon dioxide, an abundant greenhouse gas, is very difficult to use as a carbon source for carbon-carbon bond formation. CO2 is highly stable and unreactive, requiring prohibitively difficult or toxic reagents in order to form carbon bonds.
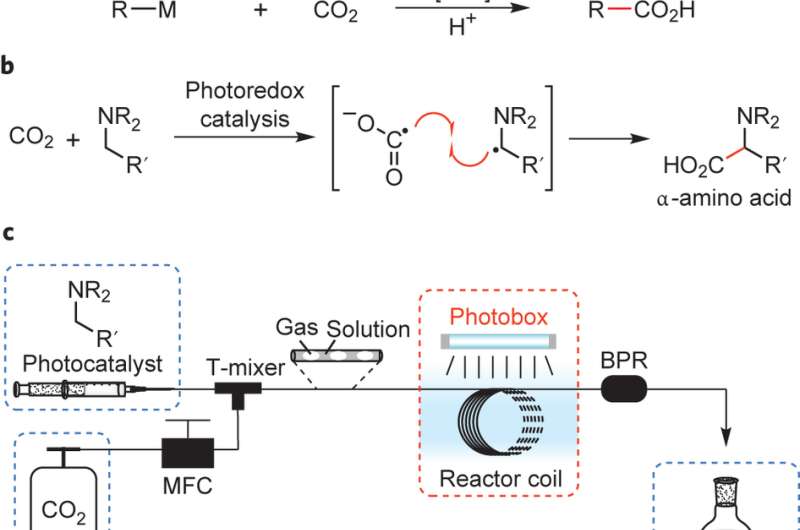
Hyowon Seo, Matthew H. Katcher, and Timothy F. Jamison of Massachusetts Institute of Technology have developed a continuous flow system that allows for the single-electron reduction of CO2 that combines with a variety of amines to form α-amino acids. Their methodology involves the use of a photoredox catalyst and, using their flow system, optimized reaction conditions to obtain regioselective α-amino acids in high yields. Their work appears in Nature Chemistry.
The keys to this group's procedure are the photoredox catalyst and the ability to fine-tune their reaction conditions using their continuous flow system. In determining the appropriate photoredox catalyst, Seo et al. tested para-terphenyl based on prior work by Yanagida's group. This catalyst was attractive because it has an appropriately high reduction potential to reduce CO2.
The photoredox catalyst served to remove an electron from the amine as well as to catalyze the reduction of CO2. These two radical molecules then combined to form a C-C bond at the benzylic C-H of an N-benzyl compound resulting in an α-amino acid. Additional experiments showed that this reaction worked well with a variety of amines including those that do not have a benzylic carbon.
The second important aspect to this reaction mechanism is the continuous flow set up. Carbon dioxide gas is mixed with a liquid aspiration of amine and catalyst. This system allowed for the controlled flow of CO2, which in turn allowed for the controlled combination of the amine radical and the CO2 radical. Seo et al. were able to optimize the pressure and flow of CO2 to obtain maximum yields. An additional advantage to this set up was the short path length of light, which also helped control when it would activate the catalyst and prompt the redox reaction to occur.
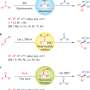
Using N-benzylpiperdine as their model reaction, Seo et al. tested their apparatus first using the reaction conditions reported by Yanagida's group. They then optimized the conditions to eventually obtain the desired α-amino acid in 92% yield. Among their changes, they added potassium trifluoroacetate (KOCOCF3) base, which helped with yields and regioselectivity. While mechanistic studies did not reveal exactly how the base is used, Seo et al. hypothesized that it served as a salt stabilizer.
Additionally, Seo et al. optimized their reaction using a UV filter. This prohibited the formation of unwanted by-products that resulted from short wave irradiation. In do this, they obtained 92% yield of the desired α-amino acid. Prior to using the UV filter, they would obtain two different regio isomers, but after the filter, they saw almost exclusive regioselectivity for the desired product (i.e., carboxylation at the benzylic position).
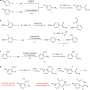
Once they obtained the optimal reaction conditions, they then tried to make a variety of α-amino acids. Notably, all cases showed a greater than 20-to-1 regioselectivity in favor of the desired α-amino acid. Among their results, they found that N-benzylpiperdines with ortho-, meta-, and para-alkyl substitution worked well, resulting in the desired α-amino acids as did chloroarenes and a variety of amines. Amines with several types of heterocycles and fused rings tolerated the reaction conditions well.
N-benzyl amines with electron poor arenes did not do as well, and neither did alcohols, ketals, or primary amines. However, masked versions of alcohols and ketals can be produced, and the reaction does tolerate 4-piperidone analogs as a possible bis-protecting group for primary amines. Also their reaction worked well when tested on a known pharmaceutical ingredient with a heterocycle and no benzylic C-H bond (ticlopidine).
"Very interesting to us is the fundamental chemistry of the radical anion of carbon dioxide, which we've proposed as a key intermediate in this process," says Professor Timothy Jamison. "Some of our ongoing efforts are directed toward using this enabling platform as a means toward this aim."
More information: Hyowon Seo et al. Photoredox activation of carbon dioxide for amino acid synthesis in continuous flow, Nature Chemistry (2016). DOI: 10.1038/nchem.2690
Abstract
Although carbon dioxide (CO2) is highly abundant, its low reactivity has limited its use in chemical synthesis. In particular, methods for carbon–carbon bond formation generally rely on two-electron mechanisms for CO2 activation and require highly activated reaction partners. Alternatively, radical pathways accessed via photoredox catalysis could provide new reactivity under milder conditions. Here we demonstrate the direct coupling of CO2 and amines via the single-electron reduction of CO2 for the photoredox-catalysed continuous flow synthesis of α-amino acids. By leveraging the advantages of utilizing gases and photochemistry in flow, a commercially available organic photoredox catalyst effects the selective α-carboxylation of amines that bear various functional groups and heterocycles. The preliminary mechanistic studies support CO2 activation and carbon–carbon bond formation via single-electron pathways, and we expect that this strategy will inspire new perspectives on using this feedstock chemical in organic synthesis.
Journal information: Nature Chemistry
© 2017 Phys.org