December 23, 2016 report
Carbanion analogs derived from naturally-occurring aldehydes

(Phys.org)—Researchers from McGill University in Montreal have devised a novel carbon-carbon bond-forming reaction that serves as an alternative to a nucleophilic addition reaction of an organometallic compound to a carbonyl (e.g., Grignard reaction). Common aldehydes, when reacted with hydrazine to form hydrazones, are then activated with a ruthenium-based catalyst and serve as the nucleophilic carbanions that attack an electrophilic carbonyl, forming secondary or tertiary alcohols. This alternative reaction demonstrated both selectivity and versatility in a variety of carbonyl compounds, including those that would serve as naturally-occurring feedstocks. Their work appears in Nature Chemistry.
"This chemistry uses catalytic amount of metal and naturally prevailing (or readily available) carbonyl functionalities to synthesize alcohols," author and project director Professor Chao-Jun Li told Phys.org.
"On the contrary, classical organometallic reagents require excessive amount of metals and pre-synthesized organohalides."
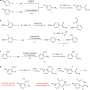
There are three components in the novel reaction reported here: the nucleophile, the electrophile, and the phosphine-based ruthenium catalyst. The nucleophile is the hydrazone, or the carbanion equivalent, formed from the aldehyde and hydrazine. The electrophile is various types of carbonyl compounds. Both the nucleophile and the electrophile can be tailored with a variety of alkyl and aryl substituents. As a model reaction, Haining Wang, Xi-Jie Dai, and Professor Li. used benzaldehyde and acetophenone to optimize the ligand and the reaction conditions.
This reaction employed umpolung reactivity of the aldehyde functional group. Umpolung reactivity means that the polarity of the carbonyl in the aldehyde is inverted from an electrophilic carbon to a nucleophilic carbon. This was done by reacting the benzaldehyde with hydrazine (N2H4) to form a hydrazone. The carbon in the C=N bond converts to a nucleophile. Using a ligand-bound ruthenium-based catalyst, a base, and a reaction additive, the hydrazone reacted with acetophenone to form a tertiary alcohol in the model reaction.
The ruthenium-based catalyst was devised by this group in previous research. It contained the ligand, 1,2-bis(dimethylphosphino)ethane. While this ligand demonstrated the best reactivity compared to other ligands, additional experiments showed that by changing the ligand, the authors could obtain a particular chiral product. Aside from the ligand, the base and the reaction additive were key ingredients for optimizing this reaction. Their preferred base was potassium phosphate, and the best additive to spurn catalytic activity was a substoichiometric amount of cesium fluoride. Using a weak base allowed the authors to try several different reactants with functional groups that could not withstand strongly basic conditions.
The authors then tested a variety of aromatic aldehydes with various functional groups to serve as the nucleophile. Many of them worked well, producing the desired alcohols in good yields. Importantly, some functional groups, such as nitriles, which are usually prohibited in Grignard reactions, performed well under these reaction conditions. Notably, there was a difference in reactivity for some substrates that was likely due to steric hindrance from the functional groups on the benzene ring. They found that sterics also played a role in the reactivity of the electrophile.
The next step was to investigate the diversity of electrophiles that could be used in this reaction. Several aromatic and aliphatic carbonyl compounds showed good reactivity and yields. The authors saw moderate-to-good yields for aromatic ketones and aldehydes, aliphatic ketones and aldehydes, and an oxo-steroid compound.
According to Professor Li "The aldehyde-derived carbanion displays excellent chemoselectivity and broad functional group tolerance, distinct from classical organometallic reagents. This is extremely important to access structurally more complex alcohols across academia and industry." The reason for this, he says, is twofold, "First, a shortened and more efficient synthetic route is possible for C-C bond formation by stitching together large pieces of carbonyl-containing molecules. Second, this allows rapid late-stage functionalization of complex molecules bearing indistinguishable functional groups other than carbonyls (e.g., esters, nitriles, alcohols, etc.)."
While the use of hydrazine precludes this reaction from being ideal for green chemistry, it does demonstrate an avenue for using naturally-occurring aldehydes that can be converted to carbanion equivalents. Furthermore, unlike other organometallic addition reactions, this reaction only required a catalytic amount of metal and produced innocuous by-products under relatively mild reaction conditions. In addition to these advantages, Professor Li points out that this procedure does not require the strict exclusion of air and water, making this an important reaction for industry.
More information: "Aldehydes as alkyl carbanion equivalents for addition to carbonyl compounds" Haining Wang, Xi-Jie Dai, and Chao-Jun Li, Nature Chemistry, DOI: 10.1038/NCHEM.2677
Abstract
Nucleophilic addition reactions of organometallic reagents to carbonyl compounds for carbon–carbon bond construction have played a pivotal role in modern chemistry. However, this reaction's reliance on petroleum-derived chemical feedstocks and a stoichiometric quantity of metal have prompted the development of many carbanion equivalents and catalytic metal alternatives. Here, we show that naturally occurring carbonyls can be used as latent alkyl carbanion equivalents for additions to carbonyl compounds, via reductive polarity reversal. Such 'umpolung' reactivity is facilitated by a ruthenium catalyst and diphosphine ligand under mild conditions, delivering synthetically valuable secondary and tertiary alcohols in up to 98% yield. The unique chemoselectivity exhibited by carbonyl-derived carbanion equivalents is demonstrated by their tolerance to protic reaction media and good functional group compatibility. Enantioenriched tertiary alcohols can also be accessed with the aid of chiral ligands, albeit with moderate stereocontrol. Such carbonyl-derived carbanion equivalents are anticipated to find broad utility in chemical bond formation.
Journal information: Nature Chemistry
© 2016 Phys.org