Researchers report in PLOS Biology the mechanism by which streptomycin, one of the oldest and most widely used antibiotics, penetrates into bacterial cells. The study performed by scientists at UT Southwestern Medical Center and their colleagues also reveals a potential way for developing new drugs to treat drug-resistant infections.
The co-lead authors are Robin Wray and Dr Irene Iscla, and co-corresponding authors are Drs Paul Blount and Junmei Wang, all from UT Southwestern Medical Center. The work was in collaboration with Dr Hua Li and Ya Gao from the School of Pharmacy at Tongji Medical College at Huazhong University of Science and Technology in China.
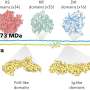
According to the World Health Organization (WHO), antibiotic and antimicrobial resistance is an increasingly serious threat to global public health. In this context, the group led by Dr Blount set out to identify compounds that would inhibit bacterial growth by altering the properties of the bacterial 'emergency release valve'—the mechanosensitive channel MscL. MscL, a transmembrane protein with a pore, is found in the vast majority of bacterial species where it helps the cell to tolerate sudden decreases in external osmolarity by releasing solutes from inside the cell. Previous studies had shown that mutations in MscL, which led to a pore that opens more easily, were detrimental to bacterial growth. Surprisingly, Dr Blount and his colleagues found that the expression of MscL increased the potency of a variant of streptomycin, dihydrostreptomycin. These results were published in Nature Communications.
Because MscL is a channel and dihydrostreptomycin increase the flux of solutes when MscL was expressed, Dr Blount and his collaboration followed the hypothesis that dihydrostreptomycin could bind and open MscL.
In the current study, using a combination of biochemical, molecular, and computational approaches, the researchers found that dihydrostreptomycin binds to a specific site of MscL and modifies its conformation, allowing the flux of solutes out of, and, surprisingly, of dihydrostreptomycin into the cell. This discovery is quite remarkable.
Streptomycin has been studied for decades, and it is firmly established that it kills bacteria mostly by interfering with protein synthesis. However, the mechanisms by which this large, bulky, and charged antibiotic accesses the inside of the bacterial cell had remained unknown until now.
Although dihydrostreptomycin channel activation is insufficient by itself to effect slowed growth or cell death in bacterial cells resistant to streptomycin's activity on protein synthesis, it is clear that the drug does directly bind and modify the MscL channel pore. In addition to solving old mysteries, the study holds promise for the discovery of new antibiotics that target the MscL channel.
Dihydrostreptomycin now serves as the first definitive example where the direct and specific binding of a compound to the MscL channel can cause at least partial, if not full opening of the MscL channel pore. If a compound could be identified that led to the more complete opening of the MscL channel or held it open for longer periods of time, it would be a worthy antibacterial candidate.
More information: Wray R, Iscla I, Gao Y, Li H, Wang J, Blount P (2016) Dihydrostreptomycin Directly Binds to, Modulates, and Passes through the MscL Channel Pore. PLoS Biol 14(6): e1002473. DOI: 10.1371/journal.pbio.1002473
Journal information: PLoS Biology , Nature Communications
Provided by Public Library of Science