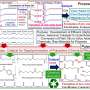
Chemical sorting can be utilized to selectively collect polyester from waste plastics and textiles. In the product stream, clean polyesters are obtained because the process eliminates colorants and most organic impurities as well. Credit: Korea Research Institute of Chemical Technology (KRICT)
The apparel industry accounts for 10% of global carbon emissions. The annual amount of fiber production reached 113 million tons in 2021 and the demand is increasing every year. However, almost 90% of post-consumer fiber wastes are disposed of through incineration or deposition in landfills.
Among these forms of waste, synthetic fiber has become a major threat to the environment and human health because, similar to other plastics, it is not biodegradable. Due to its low cost and durability, polyester is the most widely used synthetic fiber on the planet, accounting for more than half of all fabrics annually produced. Comprehensive recycling of polyester is thus a critical challenge for environmental sustainability and the health of future generations.
In practice, crude textile waste is not suitable for reuse or recycling because it is mixed with different fabric materials, colored by different dyes, and contaminated by various other impurities. Sorting it into homogeneous materials is necessary to make the waste recyclable by a chemical or mechanical method. To this end, a research team at the Korea Research Institute of Chemical Technology (KRICT) has developed a new chemical technology referred to as "chemical sorting." Their study is published in the journal ACS Sustainable Chemistry & Engineering.
This technology is applied to separate polyester from waste textiles that are disposed of in a mixed and contaminated form. In the process, a unique chemical compound, which selectively disrupts the chemical interaction between polyester and the dye used for its color, is used for the separation. The research team has also developed a new chemical recycling technology that consumes less energy than conventional methods to convert polyester into valuable monomers, which can be repeatedly used for the synthesis of polymer materials.
Postconsumer clothes, made up of various materials with unknown compositions, are often discarded. They commonly comprise a variety of textiles such as cotton, wool, polyester, acrylic, nylon, elastane, and other blended fibers. Recycling cannot be achieved without sorting them into individual materials because of their incompatible chemical and physical properties.
Industrially, the separation of individual materials from waste fabrics is accomplished by manual sorting, largely depending on human labor. This method has low accuracy and is unreliable and in turn fails to collect homogeneous materials, which is often critical for further steps of recycling. Recently, studies to develop an automatic sorting machine employ hyperspectral imaging technologies to acquire structural information of individual fabric targets. However, the sorting system still remains far from commercialization, mainly due to technical and economic barriers.
The KRICT research team adopted an inexpensive and non-toxic biodegradable compound to chemically discriminate polyester from a mixture of waste fabrics. When the compound is applied to textiles, colorants only present in polyester are completely extracted while no significant changes occur in other materials. As a consequence, clean polyester can be separated from the mixture of colored fabrics.
The method can be applied to select polyester from an uncolored fabric mixture as well. When uncolored fabric comes into contact with the waste colorants extracted from the sorting process, only polyester accepts the colorants while the other materials remain unchanged. As a consequence, the fabrics containing only polyester can be separated from mixed fabric waste in an inexpensive, accurate, and facile manner. The resulting sorted polyester can be used as clean feedstock for chemical recycling because the sorting method eliminates most organic impurities including intractable dyes.
The video describes the sequence of chemical recycling of textile wastes, containing both natural and synthetic fibers. Credit: Korea Research Institute of Chemical Technology (KRICT)
Chemical recycling, which converts polymer waste into the original building blocks, has the potential to achieve circularity in recycling of polyester wastes whereas mechanical recycling can be used to produce only low quality material. In the conventional chemical recycling method, a high reaction temperature of above 200℃ is required to completely decompose polyester. Furthermore, energy-intensive purification steps are also inevitable in most commercial applications to obtain a high quality monomer product.
The KRICT research team has developed a low-temperature glycolysis reaction system to convert chemically sorted waste polyester into pure bis(2-hydroxyethyl) terepthalate, which is an important building block monomer to produce new polymers. Monomer compounds obtained from the chemical recycling have quality equivalent to that derived from petroleum. Since the same compound as that used in "chemical sorting" functions as an additive to lower the energy barrier of depolymerization, the reaction system can be easily and economically integrated with the chemical sorting technology for applications involving plastic or textile recycling where there is high demand for good product quality.
Dr. Cho said, "Recently, the garment industry has utilized transparent and clean post-consumer PET bottles to produce recycled polyester clothes. However, this method is not sustainable because the material cannot be repeatedly recycled. In contrast, our current technology would not be limited by the complexity of the constituent materials or the initial level of impurity in the waste. Whether the targeted materials are derived from petroleum directly or recycled from waste, the technology can repeatedly process most post-consumer textile streams. Thus it will help reduce waste in landfills and substantially achieve a circular economy in the plastic and textile industries."
The chemical recycling technology has been licensed to Renew System Co., Ltd. (South Korea). Multidisciplinary R&D teams are now closely working together to build multi-scale facilities for the chemical recycling of waste clothing. A demonstration plant will be ready by the end of 2024 and commercial operation with an annual capacity of 10,000 tons is planned to start in 2025.
More information: Ngan Hong Le et al, Low-Temperature Glycolysis of Polyethylene Terephthalate, ACS Sustainable Chemistry & Engineering (2022). DOI: 10.1021/acssuschemeng.2c05570
Provided by National Research Council of Science & Technology