A high-performance catalyst that dissolves polyester and realizes chemical recycling
Professor Kotohiro Nomura's research group at Tokyo Metropolitan University developed two high-performance catalysts for efficient synthesis of value-added chemicals (fine chemicals, monomers) from polyester and vegetable oil. Their major finding is that simply heating a mixture of polyester and alcohol could convert the solution into raw materials. Their research is published in ACS Sustainable Chemistry & Engineering.
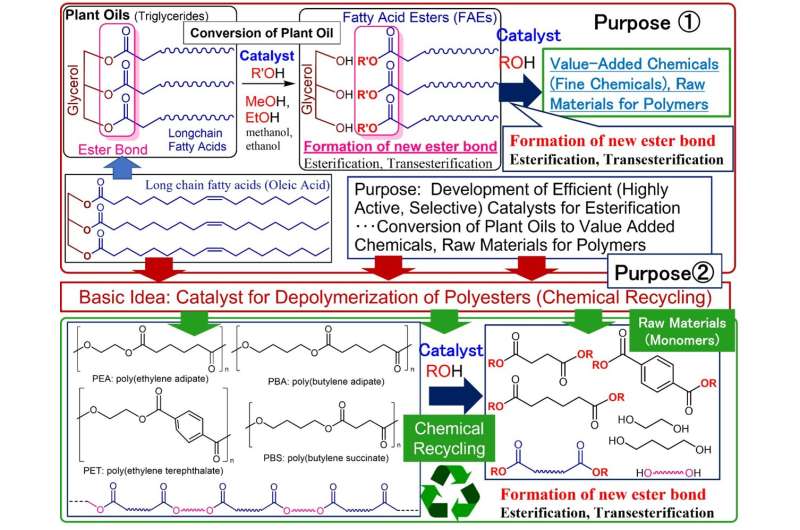
Plastic waste is an enormous environmental problem that needs to be solved immediately, but the amount of plastic being reused is still low. Polyesters, which consist of repeated "ester bonds" formed by the reaction of carboxylic acid and alcohol, are used in plastic bottles and clothing. If these ester bonds could be completely severed, polyester could be converted back into its raw materials. However, conventional methods require high temperatures and large amounts of acidic and/or basic materials. Therefore, a simple, inexpensive, and environmentally friendly method is desired.
Nomura's research group developed catalysts to facilitate the synthesis of high value-added chemicals (i.e., fine chemicals) such as the raw materials for polymers, detergents, and cosmetics from inedible vegetable oils, and discovered two types of high-performance catalysts: a calcium oxide catalyst and a titanium catalyst. These catalysts are effective for breaking down polyester based on the same chemical reaction (transesterification) and have been shown to be capable of converting polyester into raw materials with nearly 100% selectivity.
Calcium oxide, which is cheap and easy to acquire, has a proven track record in industrial applications. However, this research group's idea of applying a catalyst used for converting vegetable oil esters directly as a polyester cracking catalyst is unprecedented in research literature. Moreover, its implementation is inexpensive with a low environmental impact. Furthermore, because these titanium catalysts enabled a wide range of applications, they hold promise for producing various fine chemicals from vegetable oil, as well as for upcycling plastic waste to produce high value-added chemicals.

More information: Swetha Sudhakaran et al, CaO Catalyzed Transesterification of Ethyl 10-Undecenoate as a Model Reaction for Efficient Conversion of Plant Oils and Their Application to Depolymerization of Aliphatic Polyesters, ACS Sustainable Chemistry & Engineering (2022). DOI: 10.1021/acssuschemeng.2c04287
Provided by Japan Science and Technology Agency (JST)