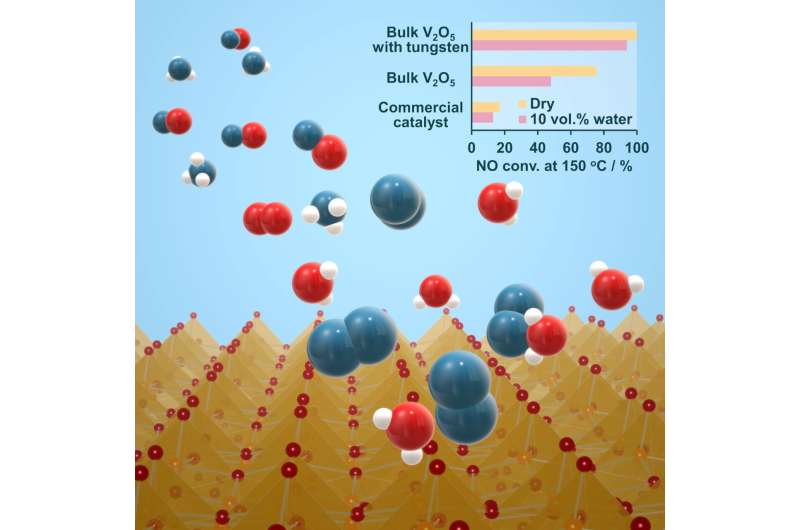
Illustration of ammonia and nitrogen oxide molecules above a crystalline catalytic material. (inset) Nitrogen oxide conversion rates at 150 degrees Celsius for tungsten-substituted bulk vanadium oxide, bulk vanadium oxide, and commercially available catalysts in both dry and "wet" conditions. Credit: Tokyo Metropolitan University
Researchers from Tokyo Metropolitan University have created a new tungsten-substituted vanadium oxide catalyst for breaking down harmful nitrogen oxides in industrial exhaust. Their new catalyst material works at lower temperatures and does not suffer major drops in performance when processing 'wet' exhaust, resolving a major drawback in conventional vanadium oxide catalysts. They found that the unaggregated dispersal of atomic tungsten in the original crystal structure plays a key role in how it functions.
Selective catalytic reduction (SCR) is an essential technology for keeping air clean. Industrial exhaust is passed through catalytic units and reacted with a reductant to convert harmful nitrogen oxides into nitrogen and water. High levels of nitrogen oxides not only harm crops and vegetation, but can directly harm people, exacerbating asthma and other respiratory problems. This makes the wide, efficient deployment of SCR technology particularly important for society.
But conventional SCR catalysts have many issues that impact performance and efficiency. For vanadium oxide catalysts which use ammonia as the reductant, a big limiting factor is performance at different temperatures. Conventional vanadium oxide catalysts work best at 200 to 400 degrees Celsius. In a boiler system, this means units have to be placed close to the combustion chamber, making them more susceptible to damage from ash. In previous work, a team led by Professor Toru Murayama of Tokyo Metropolitan University created a new bulk vanadium oxide catalyst that works efficiently at 100 to 150 degrees Celsius. However, lower temperatures led to another issue—water vapor. At lower temperatures, industrial exhaust typically contains 10-20% water vapor by volume. Since vanadium oxide catalysts suffer from a severe drop in performance when the gas is wet, further breakthroughs were required to make the most of this new catalytic material.
Now, the same team have solved this issue by creating a new, tungsten-substituted bulk vanadium oxide catalyst. By replacing some of the vanadium in the crystalline structure of the catalyst with tungsten, they discovered it was no longer susceptible to large drops in performance. At 150 degrees Celsius, while the team's previous bulk vanadium oxide catalyst suffered a conversion rate drop from 82 to 47% when the gas was wet, the new tungsten-substituted catalyst's performance only dropped from >99% conversion to 94%. This makes the material ideal for processing real industrial exhaust.
However, more tungsten did not necessarily mean better performance. The team found that the unaggregated atomic dispersion of the right amount of tungsten was vital. Both tungsten and vanadium need to work together: in wet conditions, the tungsten-substituted material made more sites available for the production of ammonium ions which could then react with nitrogen oxides near neighboring vanadium ions. The team hope their insights into the mechanism and the superior performance of their catalyst under realistic conditions will lead to new industrial SCR products and cleaner air in the not-too-distant future.
More information: Yusuke Inomata et al, Bulk tungsten-substituted vanadium oxide for low-temperature NOx removal in the presence of water, Nature Communications (2021). DOI: 10.1038/s41467-020-20867-w
Journal information: Nature Communications
Provided by Tokyo Metropolitan University