This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Chemical rope trick at molecular level: Mechanism research helps when 'trial and error' fails
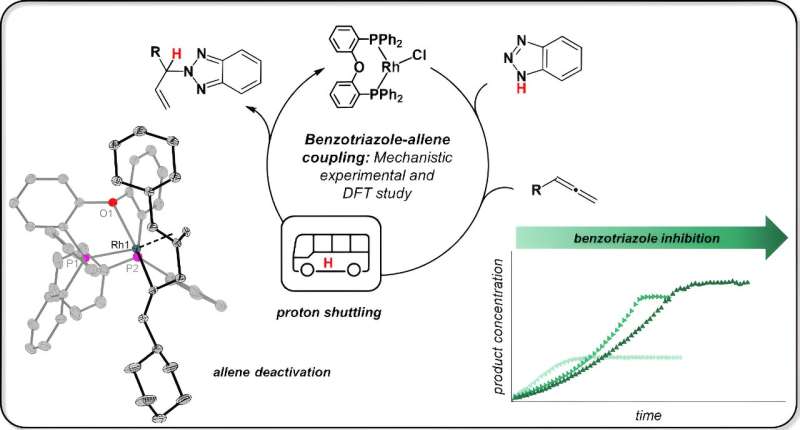
In most industrial chemical reactions, catalysts combine with the starting materials and accompany them through intermediate stages to the product. In chemistry, this pathway is known as the reaction mechanism, and it is a kind of black box: nobody knows what is happening at the molecular level at first.
If the reaction result in the laboratory falls short of expectations, chemists first resort to trial and error. Put simply, they modify the reaction until it works. Sometimes, however, it is worth taking a closer look at the reaction mechanism, as Dr. Nora Jannsen at the Leibniz Institute for Catalysis in Rostock shows using a model reaction.
She recently published her findings, which she obtained as part of her doctorate, in the Journal of the American Chemical Society.
The model reaction sounds unspectacular and, above all, strange to the layman's ear, explains Jannsen. "Benzotriazole, usually a corrosion inhibitor, is converted into a new substance with an allene (the emphasis here is on the second syllable) using a rhodium catalyst." This is then called allylbenzotriazole and can be used in a variety of syntheses. The reaction was developed by a research group led by Prof. Breit at the University of Freiburg.
Functional group set precisely
The colleagues in Freiburg succeeded in precisely linking a "functional group" to a very specific position in benzotrialzole. This is where a nitrogen–hydrogen bond (N–H) is located, as Jannsen explains.
"Functional groups" are important as molecular segments because they are responsible for the specific, e.g., pharmaceutical, effect.
"The Freiburg team wanted to place such a group in benzotriazole exactly where the N–H bond is, and the N–H bond has to give way for this," Jannsen continues. The colleagues managed to do this. "But they didn't understand on what basis they had achieved this."
That is unfortunate. Because only with an understanding of the molecular events can the reaction be specifically applied and optimized in the future. This was the aim of Jannsen's dissertation, which she successfully defended in Rostock in 2023.
Idea: Catalyst breaks bond
So how does such a reaction work? The start and end are known. What is also known about this reaction is that every single atom from the starting materials is later found in the product; chemists call this an "atom-economic" reaction. This means that in the course of the reaction, the hydrogen from the N–H bond must pass from the benzotriazole to the second starting material, the allene.
Jannsen says, "The Freiburg scientists thought that the catalyst breaks the nitrogen–hydrogen bond, i.e., a so-called oxidative addition of the benzotriazole takes place. Rhodium catalysts are known for this type of reaction."
Jannsen tested the idea by first reacting the rhodium catalyst with only one starting material, benzotriazole. She isolated samples from this reaction for nuclear magnetic resonance spectroscopy (NMR) and X-ray crystal structure analysis. The result: "The N–H bond is not broken at all. The benzotriazole remains completely intact, it only attaches itself to the catalyst."
Result: Simple coordination
The chemist then discovered that the second starting substance, allen, also binds to the catalyst. And this is where the following happens: "The two starting materials come into direct contact with each other, and the benzotriazole transfers the hydrogen atom, also known as the proton, to the allene. The catalyst merely holds the starting materials in place, but does not intervene directly in this step."
Jannsen then substantiated this proposal in detail using quantum mechanics, i.e., by theoretically modeling the reaction pathway.
Jannsen was surprised that the catalyst managed the whole thing so simply. For laypeople, it may sound a bit like a chemical rope trick. In technical terms, such a process is well known and is called protonation.
This process had an advantage. Jannsen says, "Interestingly, the reaction conditions for protonation can be significantly improved by adding another proton source." For example, she succeeded in reducing the reaction temperature from 80°C to room temperature by adding a type of co-catalyst.
A lot does not always help a lot
Jannsen also discovered that the benzotriazole can also block the catalyst under certain circumstances. This is the case when this starting substance occupies the catalyst twice, so that its reaction partner, the allen, cannot find a free space on the catalyst.
This paralyzes the catalytic activity and leads to the catalyst being "consumed." "It probably helps here to simply add the starting material to the reaction successively so that the catalyst comes into less contact with it," Jannsen says.
More information: Nora Jannsen et al, The Mechanism of Rh(I)-Catalyzed Coupling of Benzotriazoles and Allenes Revisited: Substrate Inhibition, Proton Shuttling, and the Role of Cationic vs Neutral Species, Journal of the American Chemical Society (2024). DOI: 10.1021/jacs.4c02679
Journal information: Journal of the American Chemical Society
Provided by Leibniz Institute for Catalysis