This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Study reveals molecular mechanisms of somatostatin receptor 5 activation by neuropeptides and drugs
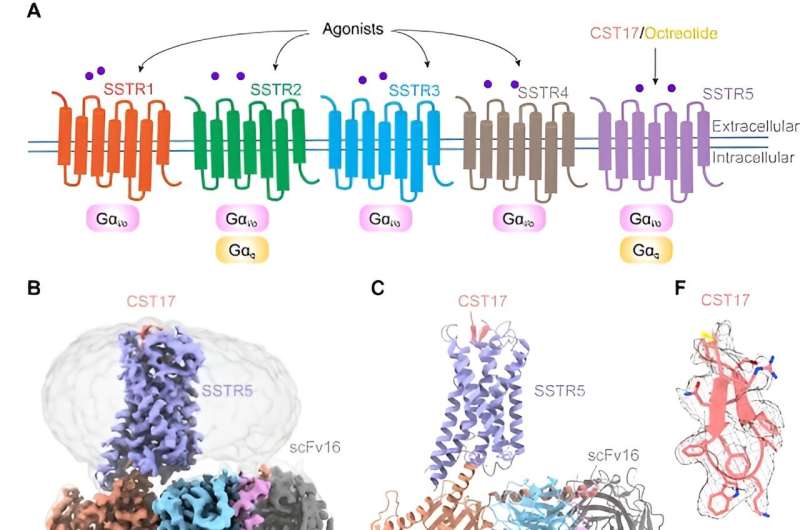
Somatostatin receptors (SSTRs) constitute a crucial family of G protein-coupled receptors (GPCRs) that play pivotal roles in regulating hormone secretion and inhibiting tumor growth.
Among five subtypes (SSTR1-SSTR5), SSTR5 stands out as a highly expressed receptor in the pituitary gland, governing the release of hormones such as adrenocorticotropic hormone, prolactin, and growth hormone. Consequently, SSTR5 emerges as a promising therapeutic target for treating endocrine disorders and tumors associated with hormonal imbalances.
In a study published in PNAS, a research team led by Eric H. Xu (Xu Huaqiang) and Zhao Lihua from the Shanghai Institute of Materia Medica (SIMM) of the Chinese Academy of Sciences, employing single-particle cryo-electron microscopy techniques, unveiled three-dimensional structures of SSTR5 in complex with the natural neuropeptide agonist cortistatin-17 (CST17) and clinically-approved drug octreotide, which sheds light on molecular mechanisms underlying SSTR5 activation by these ligands.
Researchers meticulously determined cryo-EM structures of SSTR5 bound to CST17 and octreotide at resolutions of 2.7 angstrom and 2.9 angstrom, respectively.
These structures revealed that the binding of these agonists triggers a rearrangement of a "hydrophobic lock" formed by the transmembrane helices TM3 and TM6, leading to an outward movement of TM6. This conformational change facilitates the coupling and activation of G protein, initiating downstream signaling cascades.
Remarkably, structural and functional analyses unveiled distinct recognition modes of extracellular loops ECL2 and ECL3 for CST17 and octreotide, providing insights into their agonist selectivity and specificity.
The significance of this study lies in its elucidation of the activation mechanisms of SSTR5 and its selective recognition of neuropeptide and drug agonists.
The study paves the way for structure-based design of novel, highly selective SSTR5 modulators with reduced off-target effects, and offers promising therapeutic opportunities for treating a wide range of conditions, including acromegaly, pituitary adenomas, neuroendocrine tumors, and hormonal imbalances.
More information: Jingru Li et al, Structural basis for activation of somatostatin receptor 5 by cyclic neuropeptide agonists, Proceedings of the National Academy of Sciences (2024). DOI: 10.1073/pnas.2321710121
Journal information: Proceedings of the National Academy of Sciences
Provided by Chinese Academy of Sciences