Credit: Unsplash/CC0 Public Domain
In order to adapt to the changing environment, bacteria must quickly transform extracellular information into appropriate intracellular reactions. Two component system (TCS) is the main signal transduction protein in prokaryotic cells to transform environmental stimuli into cellular responses.
HptRSA is a newly discovered TCS, which is composed of the glucose-6-phosphate (G6P) related sensor protein HptA, transmembrane histidine kinase HptS, and cytoplasmic effector HptR. It mediates G6P uptake and supports the growth and proliferation of Staphylococcus aureus, a major human pathogen, in different host cells. However, the molecular mechanism of sensing the G6P signal and triggering a downstream reaction by the HptRSA sensor complex has been a mystery.
Recently, a team led by Prof. Tao Yuyong from the School of Life Sciences, University of Science and Technology of China of the Chinese Academy of Sciences, in cooperation with Hefei National Laboratory for Physics Sciences at the Microscale, revealed the signal transduction mystery inside S. aureus, using a comprehensive application of biochemical and structural biology research methods. The study was published online in PNAS.
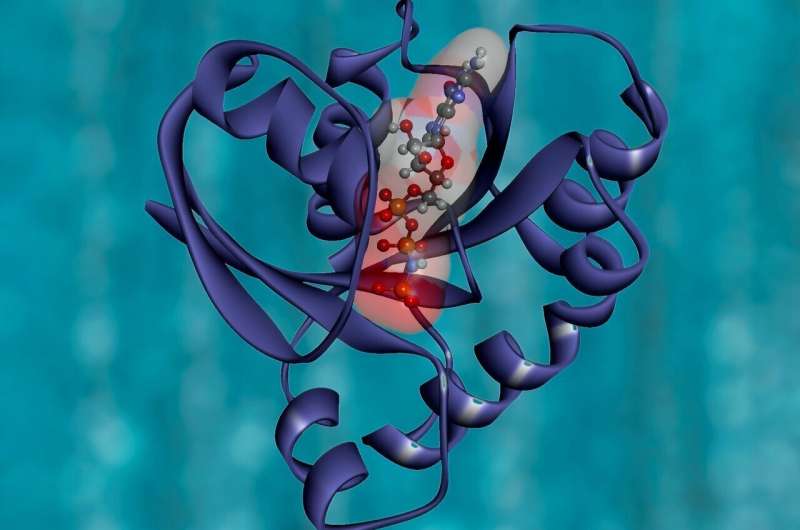
By analyzing the HptA structures in the substrate-free state and G6P binding state respectively, scientists found that G6P could bind to the gap between two HptA proteins and cause the two HptA proteins to join to each other.
The complex structures of HptA protein and HptSp shows that HptA can interact with HptS through a constitutive interface and another switchable interface. When G6P is not bound, HptA and HptSp are bound far away from the membrane and cause two HptSps to be arranged in parallel. When HptA binds to G6P, the junctions of HptA and HptSp are parallel to each other and switch to the side close to the membrane, causing the rotation of HptSp. The C-terminal of two HptSps then approach each other, transducing extracellular signal into the cell.
On the basis of the above structural discovery, scientists combined biochemical and growth analysis of HptA and HptS mutants, and proposed the G6P HptRSA signal transduction mechanism mediated by an interface switch. These results provide important clues for the nutritional sensing mechanism of bacteria, and expand the understanding of TCS activation mode for external signal transmission.
More information: Mingxing Wang et al, Interface switch mediates signal transmission in a two-component system, Proceedings of the National Academy of Sciences (2020). DOI: 10.1073/pnas.1912080117
Journal information: Proceedings of the National Academy of Sciences
Provided by University of Science and Technology of China