Focal plane detector of the TASCA separator, into which the mendelium-244 isotope was implanted and its decay registered. Credit: Alexander Yakushev, GSI Helmholtzzentrum fuer Schwerionenforschung
Gaining a better understanding of the limiting factors for the existence of stable, superheavy elements is a decade-old quest of chemistry and physics. Superheavy elements, as are called the chemical elements with atomic numbers greater than 103, do not occur in nature and are produced artificially with particle accelerators. They vanish within seconds.
A team of scientists from GSI Helmholtzzentrum fuer Schwerionenforschung Darmstadt, Johannes Gutenberg University Mainz (JGU), Helmholtz Institute Mainz (HIM) and the University of Jyvaeskylae, Finland, led by Dr. Jadambaa Khuyagbaatar from GSI and HIM, has provided new insights into the fission processes in those exotic nuclei and for this, has produced the hitherto unknown nucleus mendelevium-244. The experiments were part of "FAIR Phase 0," the first stage of the FAIR experimental program. The results have now been published in the journal Physical Review Letters.
Heavy and superheavy nuclei are increasingly unstable against the fission process, in which the nucleus splits into two lighter fragments. This is due to the ever-stronger Coulomb repulsion between the large number of positively charged protons in such nuclei, and is one of the main limitations for the existence of stable superheavy nuclei.
The nuclear fission process was discovered more than 80 years ago and is being studied intensely to this day. Most experimental data on the spontaneous fission are for nuclei with even numbers of protons and neutrons—called 'even-even nuclei.' Even-even nuclei consist entirely of proton and neutron pairs and their fission properties are rather well describable by theoretical models. In nuclei with an odd number of either neutrons or protons, a hindrance of the fission process when compared to the properties of even-even nuclei has been observed and traced back to the influence of such a single, unpaired constituent in the nucleus.
However, the fission hindrance in 'odd-odd nuclei,' containing both, an odd number of protons and an odd number of neutrons, is less well known. Available experimental data indicate that the spontaneous fission process in such nuclei is greatly hindered, even more so than in nuclei with only one odd-numbered type of constituents.
Once the fission probability is most reduced, other radioactive decay modes like alpha decay or beta decay become probable. In beta decay, one proton transforms into a neutron (or vice versa) and, accordingly, odd-odd nuclei turn into even-even nuclei, which typically have a high fission probability. Accordingly, if a fission activity is observed in experiments on the production of an odd-odd nucleus, it is often difficult to identify whether fission occurred in the odd-odd nucleus, or not rather started from the even-even beta-decay daughter, which can then undergo beta-delayed fission. Recently, Dr. Jadambaa Khuyagbaatar from GSI and HIM predicted that this beta-delayed fission process may be very relevant for the heaviest nuclei and—in fact—may be one of the main decay modes of beta-decaying superheavy nuclei.
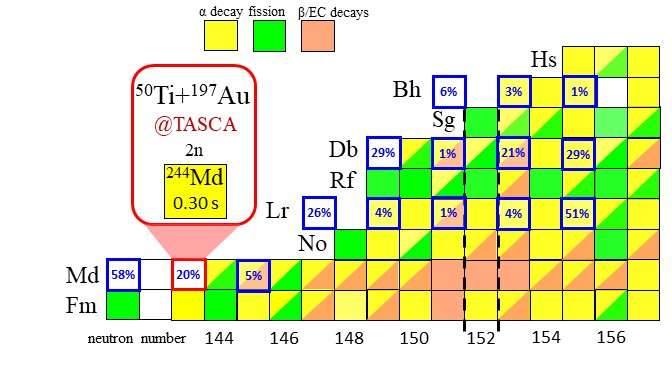
Cut out of the chart of nuclei in the region of the mendelevium nuclei. Each box represents an atomic nucleus, with the numbers of protons increasing in the vertical direction and the numbers of neutrons in the horizontal direction. Known nuclei are shown by colored boxes, where the color indicates the nuclear decay mode: alpha decay (yellow), beta decay (brown), spontaneous fission (green). Thick-framed boxes indicate odd-odd nuclei, in which beta-delayed fission has been predicted to occur with >1 % probability among all beta decays (data taken from J. Khuyagbaatar, Eur. Phys. J. A 55, 134 (2019)). The probabilities are indicated in blue. The location and decay properties of the new isotope mendelevium-244 are highlighted. Credit: J. Khuyagbaatar, GSI Helmholtzzentrum für Schwerionenforschung
In superheavy nuclei, which are exceedingly difficult to be produced experimentally, beta-decay has not yet been observed conclusively. For instance, in the case of the heaviest element produced at GSI Darmstadt, tennessine (element 117), only two atoms of the odd-odd nucleus tennessine-294 were observed in an experiment that lasted about one month. This small production rates limit the verification and detailed study of the beta-decay delayed fission process. Still, new experimental data to shed light on this process are best gained in exotic nuclei, like those which have an extremely unbalanced ratio of protons to neutrons. For this, the team from GSI, JGU, HIM and University of Jyväskylä has produced the hitherto unknown nucleus mendelevium-244, an odd-odd nucleus consisting of 101 protons and 143 neutrons.
The theoretical estimate suggests that beta decay of this nucleus will be followed by fission in about one out of five cases. Due to the large energy release of the fission process, this can be detected with high sensitivity, whereas beta decays are more difficult to measure. The researchers used an intense beam of titanium-50 available at GSI's UNILAC accelerator to irradiate a gold target. The reaction products of titanium and gold nuclei were separated in the Transactinide Separator and Chemistry TASCA, which guided mendelevium nuclei into a silicon detector suitable to register the implantation of the nuclei as well as their subsequent decay.
A first part of the studies, performed in 2018, led to the observation of seven atoms of mendelevium-244. In 2020, the researchers used a lower titanium-50 beam energy, which is insufficient to lead to mendelevium-244 production. Indeed, signals like those assigned to mendelevium-244 in the 2018 study were absent in this part of the data set, corroborating the proper assignment of the 2018 data and confirming the discovery of the new isotope.
The leader of the experiment, Dr. Jadambaa Khuyagbaatar standing in the experimental hall X8 in front of the TASCA separator used in the mendelevium-244 experiment. Credit: Antonio Di Nitto
All of the seven registered atomic nuclei underwent alpha decay, i.e., the emission of a helium-4 nucleus, which led to the daughter isotope einsteinium-240, discovered four years ago by a preceding experiment carried out at the University of Jyväskylä. Beta decay was not observed, which allows establishing an upper limit on this decay mode of 14 percent. If the 20 percent fission probability of all beta-decaying nuclei were correct, the total probability for beta delayed fission would be at most 2.8 percent and its observation would necessitate the production of substantially more mendelevium-244 atoms than in this discovery experiment.
In addition to the alpha-decaying mendelevium-244, the researchers found signals of short-lived fission events with unexpected characteristics concerning their number, production probability, and half-life. Their origin cannot currently be pinpointed exactly, and is in fact not readily explicable with current knowledge of the production and decay of isotopes in the region of mendelevium-244. This motivates follow-up studies to get more detailed data, which will help shed further light on the fission process in odd-odd nuclei.
More information: J. Khuyagbaatar et al. Search for Electron-Capture Delayed Fission in the New Isotope Md244, Physical Review Letters (2020). DOI: 10.1103/PhysRevLett.125.142504
Journal information: Physical Review Letters
Provided by Helmholtz Association of German Research Centres