Credit: Xue Liu
Leiden chemists have created a new ultrathin membrane only one molecule thick. The membrane can produce a hundred times more power from seawater than the best membranes used today. The researchers have published their findings in Nature Nanotechnology.
Thin and porous
When fresh and saltwater meet, an exchange of salt and other particles takes place. A membrane placed in water is able to harness energy from particles moving from one side to the other. A similar process can also be used to desalinate seawater. Leiden chemists have developed a new membrane that can produce a hundred times more energy than classic membranes and known prototype membranes in scientific literature.
How much power is generated depends on the thickness of the membrane and how porous it is. Researchers were able to create a carbon based membrane that is both porous and thin. That is why it can produce more energy than current membranes, which are either porous or thin, but not both.
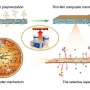
To create this new membrane, Xue Liu and Grégory Schneider spread a large number of oily molecules on a water surface. These molecular building blocks then form a thin film on their own. By heating the film, the molecules are locked in place, creating a stable and porous membrane. According to Xue Liu, the membrane can be adapted for specific requirements. Liu: "The membrane we've created is only two nanometers thick and permeable to potassium ions. We can change the properties of the membrane by using a different molecular building block. That way we can adapt it to suit any need."
Graphene
The new carbon membrane is similar to graphene, a large flat membrane made up of only carbon atoms. But according to Grégory Schneider, this new membrane is in a whole different category. Schneider: "When making a membrane, a lot of researchers start out with graphene, which is very thin, but not porous. They then try to punch holes in it to make more permeable. We've done the reverse by assembling small molecules and building a larger porous membrane from those molecules. Compared to graphene, it contains imperfections, but that's what gives it its special properties."
This new membrane combines the best of both worlds. Schneider: "Much of the research in this field was focused on creating better catalysts, membranes were somewhat of a dead end. This new discovery opens up whole new possibilities for power generation, desalination and for building much more efficient fuel cells."
More information: Xue Liu et al. Power generation by reverse electrodialysis in a single-layer nanoporous membrane made from core–rim polycyclic aromatic hydrocarbons, Nature Nanotechnology (2020). DOI: 10.1038/s41565-020-0641-5
Journal information: Nature Nanotechnology
Provided by Leiden University