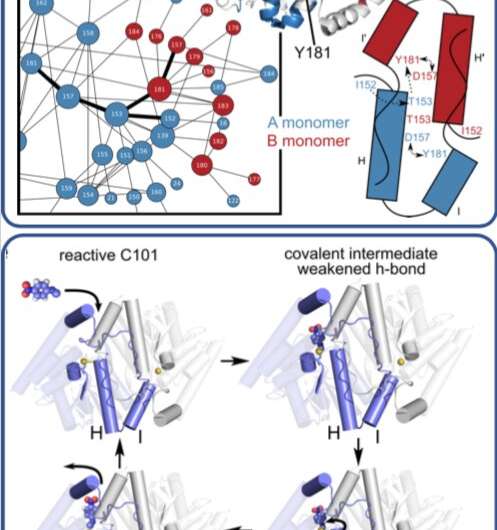
This illustration shows how an enzyme moves and changes as it catalyzes complex reactions and breaks down organic compounds. Credit: Proceedings of the National Academy of Sciences
In a time-resolved X-ray experiment, researchers uncovered, at atomic resolution and in real time, the previously unknown way that a microbial enzyme breaks down organic compounds.
The team, led by Mark Wilson at the University of Nebraska Lincoln (UNL) and Henry van den Bedem at the Department of Energy's SLAC National Accelerator Laboratory (now at Atomwise Inc.), published their findings last week in the Proceedings of the National Academy of Sciences. What they learned about this enzyme, whose structure is similar to one that is implicated in neurodegenerative diseases such as Parkinson's, could lead to a better understanding of how antibiotics are broken down by microbes and to the development of more effective drugs.
Previously, the researchers used SLAC's Stanford Synchrotron Radiation Lightsource (SSRL) to obtain the structure of the enzyme at very low temperatures using X-ray crystallography. In this study, Medhanjali Dasgupta, a UNL graduate student who was the study's first author, used the Linac Coherent Light Source (LCLS), SLAC's X-ray laser, to watch the enzyme and its substrate within the crystal move and change as it went through a full catalytic cycle at room temperature.
The scientists used special software, designed by van den Bedem, that is highly sensitive to identifying protein movement from X-ray crystallography data to interpret the results, revealing never-before-seen motions that play a key role in catalyzing complex reactions, such as breaking down antibiotics. Next, the researchers hope to use LCLS to obtain room temperature structures of other enzymes to get a better look at how the motions occurring within them help move along reactions.
More information: Medhanjali Dasgupta et al. Mix-and-inject XFEL crystallography reveals gated conformational dynamics during enzyme catalysis, Proceedings of the National Academy of Sciences (2019). DOI: 10.1073/pnas.1901864116
Journal information: Proceedings of the National Academy of Sciences
Provided by Stanford University