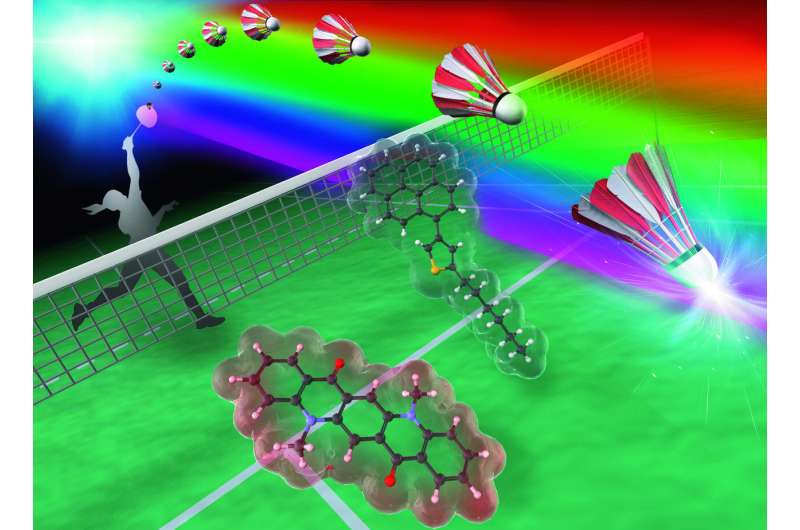
The two-component dye shows self-recovering mechanochromic luminescence that exhibits a high-contrast emission color change between violet and orange. Credit: Yokohama National University
The building blocks of rationally designed chemicals are simple elements: carbon, hydrogen, oxygen and so on. These elements can be combined in myriad ways to accomplish a variety of chemicals with different characteristics. Even the same chemical can be treated differently—with pressure or heat, for example—to show drastically different properties. A simpler version is to think of how water can be boiled to cook pasta or frozen to become ice—the same ingredient can be made into two different states via temperature treatment.
Now, researchers are working to better control how the chemicals respond to treatment, as well as how to reverse the chemicals back to their original state with little to no interference. Such control would allow scientists to prepare the sensing systems of environmental stimuli, as well as continuously repeat the sensing.
A team of researchers at Yokohama National University has achieved such results with a specific compound that can emit light and has potential applications in the next generation of smart devices such as wearable devices and anti-counterfeiting paintings. They published their results online on September 12, ahead of print in Chemical Communications.
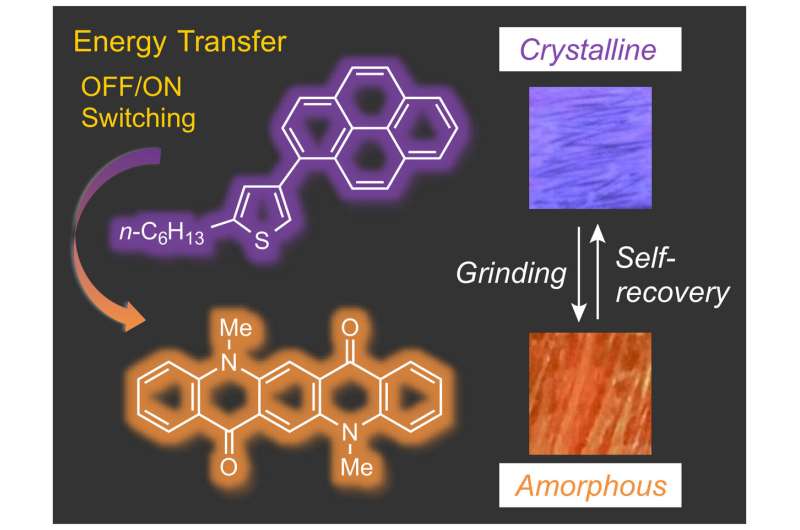
The compound is a derivative of thiophene, which is a dye with mechanochromic luminescence properties—it changes color under physical change. It starts emitting a violet glow under the irradiation of UV light, but as it is exposed to mechanical stimuli, such as grinding, the violet glow shifts slightly to blue. Another external intervention can make the compound heal and become violet again.
"Mechanochromically luminescent (MCL) dyes have recently attracted considerable interest on account of their potential applications," said Suguru Ito, paper author and associate professor in the Department of Chemistry and Life Science in the Graduate School of Engineering Science at Yokohama National University. "However, it is still very difficult to rationally design MCL dyes with desired characteristics."
The two-component dye shows self-recovering mechanochromic luminescence that exhibits a high-contrast emission color change between violet and orange. Credit: Yokohama National University
In this study, however, researchers discovered that by adding another chemical called DMQA, the dye changed to orange under mechanical stimuli. The dye did not need more external stimuli to revert back to violet either.
"We combined two kinds of rational design guidelines for tuning the luminescent properties, resulting in the desired—and unprecedented— characteristics of high-contrast, self-recovering dyes," Ito said.
The first rational design guideline is that the recovery behavior of the dye can be attributed to the length of the alkyl group in the compound—a longer chain of carbon atoms with hydrogens in the dye allows the dye to recrystallize and heal in time. The second is that by mixing with DMQA, the color range between the original state and ground state differ greatly.
"The next step is to establish a rational design guideline to control the dye's responsiveness to mechanical stimuli," Ito said. "My ultimate goal is to develop an innovative pressure-sensing system by rationally creating a material that can change its emission color in stages in response to mechanical stimuli of different intensity."
With such control, Ito could use mechanical stimuli to precisely induce a specific and intended response. A little pressure could shift the violet glow to blue, a little more pressure pushes the glow closer to red. A system with such ability would allow for stepwise changes and recoveries by the stimulus, which could be highly beneficial in the next generation of smart materials, according to Ito.
More information: Minako Ikeya et al, Tunable mechanochromic luminescence of 2-alkyl-4-(pyren-1-yl)thiophenes: controlling the self-recovering properties and the range of chromism, Chemical Communications (2019). DOI: 10.1039/C9CC06406K
Journal information: Chemical Communications
Provided by Yokohama National University