Fluoridating two-dimensional tungsten disulfide adds metallic islands to the synthetic semiconductor, along with unique optical and magnetic properties, according to researchers at Rice University. Credit: Ajayan Research Group/Rice University
By getting in the way, fluorine atoms help a two-dimensional material transform from a semiconductor to a metal in a way that could be highly useful for electronics and other applications.
A study led by Rice materials scientist Pulickel Ajayan and lead author Sruthi Radhakrishnan details a new method to transform tungsten disulfide from a semiconductor to a metallic state.
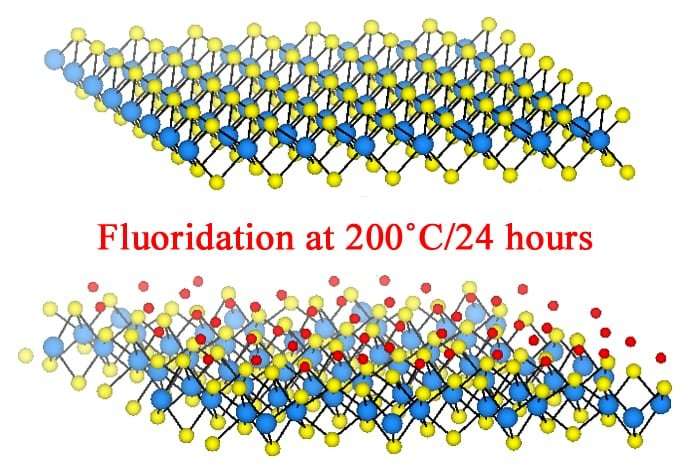
Other labs have achieved the transformation by adding elements to the material – a process known as doping – but the change has never before been stable. Tests and calculations at Rice showed fluorinating tungsten disulfide locks in the new state, which has unique optical and magnetic properties.
The researchers also noted the transformation's effect on the material's tribological properties—a measure of friction, lubrication and wear. In short, adding fluorine makes the material more slippery at room temperature.
The lab's work is detailed in Advanced Materials.
Tungsten disulfide is a transition metal dichalcogenide (TMD), an atom-thick semiconductor. Unlike graphene, which is a flat lattice of carbon atoms, a TMD incorporates two elements, one a transition metal atom (in this case, tungsten) and the other (sulfur) a chalcogen. The material isn't strictly flat; the transition metal layer is sandwiched between the chalcogen, forming a three-layered lattice.
TMDs are potential building blocks with other 2-D materials for energy storage, electrocatalysis and lubrication, all of which are influenced by the now-stable phase transformation.
Fluoridating two-dimensional tungsten disulfide adds metallic islands to the synthetic semiconductor, along with unique optical and magnetic properties, according to researchers at Rice University. Credit: Ajayan Research Group/Rice University
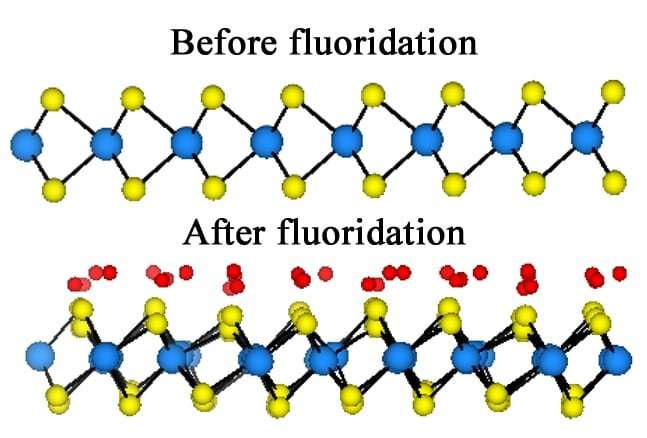
Because fluorine atoms are much smaller than the 0.6-nanometer space between the layers of tungsten and sulfur, the researchers said the invasive atoms work their way in between, disrupting the material's orderly lattice. The fluorine allows the sulfur planes to glide this way or that, and the resulting trade of electrons between the fluorine and sulfur also accounts for the unique properties.
"It was certainly a big surprise. When we started this work, a phase transformation was the last thing we expected to see." said Radhakrishnan, a former graduate student in Ajayan's lab and now a module engineer at Intel Corp. in Hillsboro, Ore.
"It is really surprising that the frictional characteristics of fluorinated tungsten disulfide are entirely different from the fluorinated graphene that was studied before," said co-author Tobin Filleter, an associate professor of mechanical engineering at the University of Toronto. "This is a motivation to study similar 2-D materials to explore such interesting behavior."
The researchers said fluorine appears to not only decrease the bandgap and make the material more conductive but also causes defects that create metallic "islands" along the material's surface that also display paramagnetic and ferromagnetic properties. "These regions of metallic tungsten disulfide are magnetic and they interfere with each other, creating interesting magnetic properties," Radhakrishnan said.
Further, because fluorine atoms are electrically negative, they're also suspected of changing the electron density of neighboring atoms. That changes the material's optical properties, making it a candidate for sensing and catalysis applications. Radhakrishnan suggested the materials may also be useful in their metallic phase as electrodes for supercapacitors and other energy-storage applications.
Radhakrishnan said different concentrations of fluorine alter the proportion of change to the metallic phase, but the change remained stable in all three concentrations the lab studied.
"The phase transformation, change in properties with functionalization by fluorine and its magnetic and tribological changes are very exciting," Ajayan said. "This can be extended to other 2-D layered materials and I am sure it will open up some captivating applications."
More information: Sruthi Radhakrishnan et al. An Insight into the Phase Transformation of WS2 upon Fluorination, Advanced Materials (2018). DOI: 10.1002/adma.201803366
Journal information: Advanced Materials
Provided by Rice University