Credit: AI-generated image (disclaimer)
Bread is often called the staff of life, but that label might be more accurately applied to nitrogen, the element that soil bacteria pluck from the atmosphere and chemically alter to help spur the growth of plants, which ultimately nourish livestock and humans as well.
Today a vast industry exists to produce and deliver nitrogen-based fertilizers to farms, which benefit from higher crop yields but, unfortunately, at some environmental cost, as excess chemical runoff often spills into rivers and coastal waterways.
Now Stanford researchers are leading a multi-year effort to produce this vital growth booster in a sustainable way, by inventing a solar-powered chemistry technology that can make this fertilizer right on the farm and apply it directly to crops, drip-irrigation style.
"Our team is developing a fertilizer production process that can feed the world in an environmentally sustainable way," says chemical engineer Jens Norskov, director of the SUNCAT Center for Interface Science and Catalysis, a partnership between researchers from Stanford Engineering and the SLAC National Accelerator Laboratory.
This eight-year SUNCAT project is supported by a $7 million grant from the Villum Foundation, an international scientific and environmental philanthropy. The sustainable nitrogen effort is part of a broader, $20 million Villum-backed initiative to bring Stanford researchers together with Danish scientists to develop sustainable technologies to produce not just fertilizers, but fuels and other vital industrial chemicals.
"One common thread across these projects is the need to identify catalysts that can promote chemical processes powered by sunlight, instead of relying on the fossil fuels now commonly used as energy sources and, often, as feedstock for reactions," says Norskov, a professor of chemical engineering and of photon science at Stanford.
Catalysts – compounds that spur reactions without being consumed – have been used on an industrial scale for more than a century. Today's fertilizers are commonly derived from petrochemicals through an energy-intensive process that relies on catalysts to accelerate reactions that occur under high pressures and temperatures. Developing a low-energy, solar-based process to make nitrogen fertilizers could benefit billions of people, particularly those in the developing world. But to get there SUNCAT researchers will have to break ground in the science of catalysis.
"We know of no manmade catalysts that can do what we require," Norskov says. "We will have to design them."
Nitrogen and life
Nitrogen is literally woven into the fabric of life. Through chemical combinations with carbon, hydrogen and oxygen, nitrogen helps form amino acids, which are themselves the building blocks of proteins, that versatile family of molecules vital to every living thing. We can thank soil bacteria for making nitrogen usable. Over time microorganisms evolved a biochemical ecosystem to extract nitrogen from the atmosphere and combine it with hydrogen from water to form compounds such as ammonia that can be absorbed by plants, promoting their growth and channeling this atmospheric gas to the food chain.
We don't know when farmers first discovered the benefits of fertilization but the practice is ancient. Modern studies of the soils around Neolithic settlements suggest that, as early as 6,000 years ago, farmers sought to boost yields by fertilizing crops with animal waste – now known to contain nitrogen-rich urea (ammonia plus carbon). Other traditional fertilization practices have included growing crops such as clover and alfalfa that are good at fixing usable nitrogen into the soil, or simply letting fields lie fallow to let soil bacteria replenish nature's supply. Over time, as population grew and moved to cities, industries arose to supply farmers with nitrogen-based fertilizers. At times this involved sending ships to scoop bird guano deposits off remote islands, or mining chemicals like sodium nitrate or ammonium sulfate that could be refined into plant growth additives.
By the first decade of the 20th century, however, population growth threatened to overtax such practices. It was at this crucial juncture that German chemist Fritz Haber, working with chemical engineer Carl Bosch, discovered how to mass produce ammonia in giant vats using natural gas, which was the starting point or feedstock of the process. Under extreme pressure and heat, chemical catalysts could crack natural gas molecules, liberating the hydrogen atoms and joining them to nitrogen from air to form NH3, or synthetic ammonia that could be readily absorbed by plants. The Haber-Bosch technology has been hailed as one of the key discoveries of the 20th century.
"We literally feed the world on fertilizers derived from the Haber-Bosch process," Norskov says.
Scale and environmental impact
Tom Jaramillo, deputy director of the SUNCAT Center and a member of the nitrogen synthesis project, put annual fertilizer production into perspective.
"Each year we produce more than 20 kilograms of ammonia per person for every person on the planet, and most of that ammonia is used for fertilizer," says Jaramillo, an associate professor of chemical engineering and of photon science at Stanford.
But this massive fertilizer output has several costs, starting with production. Due to the heat and pressure required by the Haber-Bosch process, ammonia catalysis accounts for approximately 1% of all global energy use. On top of that, between 3% and 5% of the world's natural gas is used as a feedstock to provide the hydrogen for ammonia synthesis.
Then come the environmental costs. Today's fertilizers are mass produced in centralized plants, delivered to farms and administered using mechanized spreaders. Rain and irrigation water can wash excess fertilizer into streams, rivers and coastal waterways. Accumulations of fertilizer runoff can spur the hyper growth of water-borne plants, creating a negative environmental spiral in which the plants can suffocate marine life to create "dead zones" in rivers, lakes and saltwater bays.
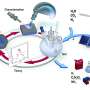
SUNCAT researchers aim to provide the benefits of fertilization without any of these costs. The idea is to replace the centralized, fossil-fuel based Haber-Bosch process with a distributed network of ammonia-on-demand production modules run off renewable energy. These modules would use solar power to pull nitrogen from the atmosphere and also to catalyze the splitting of water molecules to get hydrogen and oxygen. The catalytic processes would then unite one nitrogen atom to three hydrogen atoms to produce ammonia, with oxygen as a waste product.
"We will harness solar energy in the presence of properly designed catalysts to create ammonia right in the agricultural fields," Norskov says. "Think of it as a drip irrigation method of synthesizing ammonia, where it percolates into the roots of the crops."
This effort comes as attention is being focused on industrialized agriculture's heavy reliance on fossil fuels and the many environmental ramifications of that dependence.
"You won't need tremendous quantities of fossil fuels as an ammonia feedstock, or to drive the trucks that deliver the fertilizers or the tractors that apply it," Norskov says. "And you won't have a problem with excess application and fertilizer runoff, because virtually all the fertilizer that is produced will be consumed completely by the crops."
Such a process would have a global payoff. In the developed economies with mechanized agriculture, solar-based nitrogen catalysis would deliver fertilizers with dramatically lower environmental costs. In regions like sub-Saharan Africa, where depleted soils have stymied efforts at sustainable agriculture and reforestation, a solar-based fertilization technology could help subsistence farmers boost crop yields and alleviate hunger.
Next-generation catalysis
Developing a solar-powered technology to produce nitrogen-based fertilizers is an enormous challenge that begins with designing the necessary catalysts.
"It is remarkable how much economic and industrial activity depends on catalysis and how little this is appreciated," Norskov says.
Catalysts are chemistry's multitaskers: They must target specific molecules, break certain chemical bonds and, often, create new bonds to remake from the atomic jumble whatever end molecule is desired. It is understandably rare to find a chemical agent that can perform all this breaking and making without becoming exhausted – in this case a technical reference to the fact that a catalyst must carry out these chemical reactions without changing the atomic structure that enabled it to perform its multitasking magic in the first place.
"While the catalyst must bind strongly enough to the target molecule to do the work required, it also has to release the end product," says Stacey Bent, a professor of chemical engineering at Stanford and key member of the SUNCAT team. "We have to design catalysts that can make and break bonds with atomic precision, and we have to ensure these materials can be mass produced at the necessary scales and price points, and are durable and simple to use in the fields."
This is especially true in the case of the fertilizer-production process envisioned here, Jaramillo explains, because of the complexity of the process.
"We have to design a series of reactions to cleave the nitrogen molecule from air, separate the hydrogen from water and combine them to form ammonia, with the only input energy coming from solar power," Jaramillo says, adding, "We're really just at the beginning."
Computation, visualization, experimentation
The close working relationship between Stanford engineers and researchers at the SLAC National Accelerator Laboratory is an important part of the story.
SLAC particle accelerators and imaging technologies can capture and visualize chemical reactions at the atomic scale. That, in combination with SLAC's computational assets, will allow the SUNCAT team to use a variety of techniques, including artificial intelligence, machine learning and simulation, to identify promising materials, and then predict how slight alterations to their atomic structures might optimize them for use as catalysts.
"We plan to simulate the properties of materials that could perform the necessary reactions," says Bent, "and then come up with a short list of the best candidates for experimentalists to synthesize and test."
The magnitude of the task requires a wide range of talents. In addition to Norskov, Jaramillo and Bent, other participating Stanford researchers include chemical engineering faculty Zhenan Bao and Matteo Cargnello. SLAC collaborators include Thomas Bligaard, senior staff scientist and deputy director of theory at SUNCAT, and staff scientist Frank Abild-Pedersen. A group of Danish researchers led by professor Ib Chorkendorff at the Technical University of Denmark are key members of the project.
"We are part of a very strong team, attacking some of the biggest challenges in chemistry, chemical engineering and sustainability," says Jaramillo.
The ultimate goal is to create a catalytic process that can spur the various ammonia-producing chemical reactions with no inputs other than air, water and sunlight. Moreover, these inexhaustible catalysts, and indeed every component in these ammonia-production modules, must be inexpensive to mass produce, durable in the field and easy to operate. It's a tall order but the potential payoff is huge.
"Sustainable nitrogen production will only become possible with the cross-disciplinary collaboration of people working in fields such as materials science, chemical engineering and computer science," Bent says. "It could literally change the world."
If the project's goal seems worth the effort, the same is true for its research methodology. Team-based discovery that combines theoretical insight, atomic-level visualization and computational simulation can be applied to designing other sustainable processes to create fuels and industrial chemicals, as envisioned by the broader Villum initiative.
Norskov framed that broader objective against the backdrop of global warming in a recent paper co-authored with Arun Majumdar, a professor of mechanical engineering at Stanford, co-director of the Precourt Institute for Energy and former founding director of the Advanced Research Projects Agency–Energy.
In an essay for the Scientific Philanthropy Alliance, Norskov and Majumdar posit that civilization has reached the point at which the technologies that have allowed our population to grow may now threaten life's underpinnings. The essential challenge of the 21st century is to develop new technologies that meet human needs in ways that are environmentally sustainable.
"Essentially we are attempting to restore the balance in the Earth's carbon and nitrogen cycles that has been lost through the exponential increase in the demand for food and fossil fuels," Norskov and Majumdar write, adding, "The time to act is now."
More information: A. Bogaard et al. Crop manuring and intensive land management by Europe's first farmers, Proceedings of the National Academy of Sciences (2013). DOI: 10.1073/pnas.1305918110
Journal information: Proceedings of the National Academy of Sciences
Provided by SLAC National Accelerator Laboratory