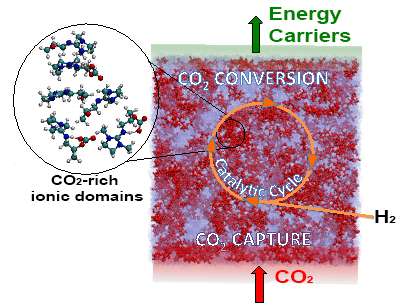
Captured carbon dioxide (CO2) can more easily undergo the catalyst-driven conversion to valuable chemicals, including energy carriers. Credit: David Heldebrant, PNNL
To convert carbon dioxide captured at coal-fired power plants into valuable chemicals requires an energy-intense exchange between the carbon being a gas or trapped in liquid. Avoiding the exchange could reduce the amount of energy used. The challenge is in understanding how the carbon dioxide acts in a liquid state. The behavior of the gaseous state is well known. Scientists at Pacific Northwest National Laboratory (PNNL) showed that carbon dioxide in solution is reactive and concentrated, far different from the stable and diffuse gaseous state.
Transportation fuels, asphalt, plastics, and other products are made from carbon atoms taken from fossil fuels. Captured carbon dioxide could replace fossil fuels as a source for some of these carbon atoms. This study uncovers key characteristics of captured pollutant that may make using it as a feedstock easier. "Combining energy-intense separation and conversion steps saves energy," said Dr. David Heldebrant, the green chemist who led the study.
Using resources at the national lab, the team at PNNL examined the behavior of the carbon dioxide atoms in solution. The negatively charged carbon compounds (a.k.a. alkylcarbonates) are reactive, potentially requiring less energy to produce chemicals or fuels such as methanol. They also found that the carbon dioxide forms into tiny balls, or micelles, in solution. These micelles are easy to concentrate carbon dioxide, whereas the gas requires high pressures.
"In a way, we are changing the carbon dioxide itself to improve the reaction," said Heldebrant.
This research is part of a broader project, led by Heldebrant, to examine capture and conversion. "If you have to pay energy to capture carbon dioxide, why not make it into something valuable-offset some of the energy cost by creating polymers, fuels, and specialty chemicals," said Heldebrant.
The team is delving into the nuances of how carbon dioxide behaves in solution, including how it moves, sticks, and reacts. Also, they are mapping vital steps in the journey from capture to fuel.
More information: D. B. Lao et al. The steps of activating a prospective COhydrogenation catalyst with combined COcapture and reduction, Green Chem. (2016). DOI: 10.1039/c6gc01800a
Provided by Pacific Northwest National Laboratory