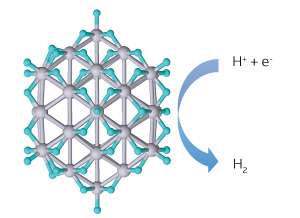
First-principles simulations reveal distribution of absorbed hydrogen atoms (cyan) on a small platinum nanoparticle catalyst during hydrogen evolution reaction. Credit: American Chemical Society
A*STAR scientists have used first-principles computer simulations to explain why small platinum nanoparticles are less effective catalysts than larger ones.
Platinum nanoparticles are used in the catalysis of many reactions, including the important hydrogen evolution reaction used in fuel cells and for separating water into oxygen and hydrogen. Improved effectiveness of platinum nanoparticles to catalyze this reaction had been experimentally shown with decreasing nanoparticle size until it fell below about 3 nanometers. There was no clear explanation for why catalytic activity was reduced at this scale.
Teck Leong Tan and colleagues at the A*STAR Institute of High Performance Computing, and collaborators at the Ames Laboratory in the USA, performed first-principles calculations of platinum nanoparticles for the hydrogen evolution reaction. Based on these calculations, they produced a map of the intermediate compounds—in this case adsorbed hydrogen atoms—that form on the nanoparticles. They also estimated the contribution made by each catalytic active species to the overall activity.
An effective catalyst must not bind to reaction intermediates too weakly because reactants will fail to bind to its surface. Too strong an adherence will cause difficulty for reaction products to detach from the catalyst surface. The binding energy of an effective catalyst should be 'just right', lying somewhere between these two extremes.
The researchers found that edge sites on small platinum nanoparticles bind too strongly to hydrogen atoms and become inactive catalytically, but face sites continue to bind with hydrogen with an appropriate energy level and remain catalytically active. The increased ratio of edge sites to face sites as nanoparticle size reduces explains the observed fall in catalytic activity for small nanoparticles. It also suggests that the nanoparticle shape could be tailored to optimize the nanoparticle's catalytic activity.
The simulation results augur well for the potential of this technique. "Experimentalists have long been trying to visualize the structure of nanosized catalysts and the adsorbate distribution in real-time during reactions," explains Tan. "However, this is often difficult to achieve. Our first-principles computational method provides an accurate model of the catalyst structure with adsorbate coverage and thus allows researchers to visualize what is going on in catalysts during a reaction."
The computational method can be applied to nanoscale catalysts besides platinum, and the team is keen to explore its potential to predict the performance of nanoparticles of other elements.
More information: "Platinum nanoparticle during electrochemical hydrogen evolution: Adsorbate distribution, active reaction species, and size effect." ACS Catalysis 5, 2376–2383 (2015). dx.doi.org/10.1021/cs501840c
Journal information: ACS Catalysis