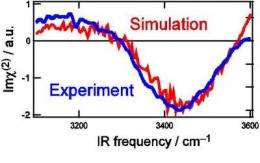
This figure shows the imaginary part of second order nonlinear susceptibility spectra of water surfaces: (a) Experimental HOD (blue line), (b) simulated HOD (red). The concentration of isotopes is H2O : HOD : D2O = 1: 6: 9. Credit: RIKEN
Findings by Japanese researchers at the RIKEN Advanced Science Institute and their colleagues at Tohoku University and in the Netherlands have resolved a long-standing debate over the structure of water molecules at the water surface. Published in the Journal of the American Chemical Society, the research combines theoretical and experimental techniques to pinpoint, for the first time, the origin of water's unique surface properties in the interaction of water pairs at the air-water interface.
The most abundant compound on the Earth's surface, water is essential to life and has shaped the course of human civilization. As perhaps the most common liquid interface, the air-water interface offers insights into the surface properties of water in everything from atmospheric and environmental chemistry, to cellular biology, to regenerative medicine. Yet despite its ubiquity, the structure of this interface has remained shrouded in mystery.
At the heart of this mystery are two broad bands in the vibrational spectrum for surface water resembling those of bulk ice and liquid water. Whether these bands are the result of hydrogen bonds themselves, of intra-molecular coupling between hydrogen bonds within a single water molecule, or of inter-molecular coupling between adjacent water molecules, is a source of heated debate. One popular but controversial hypothesis suggests one of the spectral bands corresponds to water forming an actual tetrahedral "ice-like" structure at the surface, but this interpretation raises issues of its own.
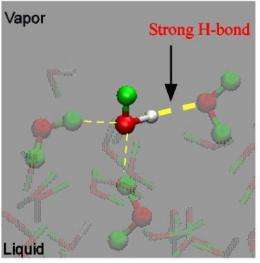
This is a snapshot in the MD simulation trajectory of the HOD / D2O mixture that shows the water pair at the surface. White, green and red represent H, D and O atoms, respectively. Credit: RIKEN
The researchers set out to resolve this debate through a comprehensive study combining theory and experiment. For their experiments, they applied a powerful spectroscopy technique developed at RIKEN to selectively pick out surface molecules and rapidly measure their spectra. To eliminate coupling effects, which are difficult to reproduce in simulations, they used water diluted with D2O (heavy water) and HOD (water with one hydrogen atom, H, replaced by deuterium, D). Doing so eliminates coupling of OH bonds within a single molecule (since there is only one OH bond) and reduces the overall concentration of OH bonds in the solution, suppressing intermolecular coupling.
With other influences removed, the researchers at last pinpointed the source of water's unique surface structure not in an "ice-like" structure, but in the strong hydrogen bonding between water pairs at the outermost surface. The extremely good match between experimental and theoretical results confirms this conclusion, at long last bringing clarity to the debate over the structure of the water surface and setting the groundwork for fundamental advances in a range of scientific fields.
Provided by RIKEN