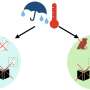
Scale on the inside of cast iron alloy pipes can react with residual disinfectant in the water to release Cr(VI). Credit: Adapted from Environmental Science & Technology 2020, DOI: 10.1021/acs.est.0c03922
The water crisis in Flint, Michigan, brought much-needed attention to the problem of potentially toxic metals being released from drinking water distribution pipes when water chemistry changes. Now, researchers reporting in ACS' Environmental Science & Technology have investigated how hexavalent chromium, known as Cr(VI), can form in drinking water when corroded cast iron pipes interact with residual disinfectant. Their findings could suggest new strategies to control Cr(VI) formation in the water supply.
The metal chromium, known as Cr(0), is found in cast iron alloy, which is the most widely used plumbing material in water distribution systems. As pipes corrode, a buildup of deposits, known as scale, forms on the pipes' inner walls. Trace chemicals in water can react with scale, forming new compounds that could be released into the water. Some of these compounds contain Cr(VI), which, at high doses, can cause lung cancer, liver damage, reproductive issues and developmental problems. In 2014, California set a drinking water standard of 10 μg/L Cr(VI), but the guideline was later withdrawn because no economically feasible treatment to remove Cr(VI) from tap water existed. Haizhou Liu and colleagues wanted to find out how exactly Cr(VI) makes its way into drinking water, which might reveal new ways to prevent its formation.
The researchers collected two sections of cast iron pipe from two drinking water distribution systems in the U.S.: one from a system using groundwater with naturally high Cr(VI) levels (11-24 μg/L), and the other from a system using surface water with undetectable Cr(VI). The team scraped off scale from the pipes and analyzed its composition. The levels of total Cr were about 18 times higher in the first pipe than in the second. In both pipes, chromium existed in two oxidation states, Cr(0) and Cr(III). When the researchers added a chlorine- or bromine-containing disinfectant to the scale, it quickly reacted with Cr(0), rather than Cr(III) as previously suspected, to form Cr(VI). To help mitigate Cr(VI) levels, adding less-reactive disinfectants to treat drinking water could be explored, and cast iron pipes with chromium alloy should be used with caution, the researchers say.
More information: "Hexavalent Chromium Release in Drinking Water Distribution Systems: New Insights into Zerovalent Chromium in Iron Corrosion Scales" Environmental Science & Technology (2020). pubs.acs.org/doi/abs/10.1021/acs.est.0c03922
Journal information: Environmental Science & Technology
Provided by American Chemical Society