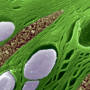
Scanning electron microscope image of thylakoids - each circle is a high-rise building seen from above. Credit: Wanner/LMU
The emergence of oxygenic photosynthesis made it possible for complex multicellular life-forms to evolve on Earth. By utilizing solar energy to turn carbon dioxide into sugars, while also generating molecular oxygen from water, photosynthesis provides the basis for both plant and animal life. These two processes are carried out by distinct, but functionally connected complexes called photosystems I and II (PSI and PSII). In cyanobacteria, algae and plants, these photosystems—all of which employ chlorophyll pigments to capture light energy—are embedded in specialized lipid membranes called thylakoids. Moreover, the thylakoids that contain PS I and PSII differ in their organization, which effectively enables the two systems to convert light of different wavelengths into chemical energy.
The functional link between the two reaction complexes is provided by soluble proteins that serve as electron transporters. Biologists led by Professor Dario Leister (Chair of Plant Molecular Biology) at LMU's Biocenter, in collaboration with international colleagues, have now taken a closer look at the role of one of these proteins—plastocyanin, a small copper-containing protein. Their findings, which appear in the journal PNAS, reveal that the efficacy of electron transport is critically dependent on the architecture of the membrane systems. "Function shapes architecture," says Leister.
This remark relates to differences in the configuration of the thylakoid membranes in which the 'light reactions' of photosynthesis (i.e. those that are driven directly by light energy) take place. Photosystem II resides in structures made up of membrane sacs, which are stacked like plates, and resemble high-rise buildings, Leister explains. These stacks are connected by unstacked thylakoids. These may be compared to enclosed walkways that run at various levels between the tower blocks. Photosystem I is located only in these walkways. To convey electrons from PSII to PSI, one therefore needs couriers—mobile electron-transport proteins. Green plants have two such transporters, plastoquinone and plastocyanin. The new study by Leister and his colleagues demonstrates that plastocyanin is the more important of the two. In addition, the experiments show that plastocyanin-mediated transport system is most efficient when the thylakoid stacks (tower blocks) are not too high and not too wide. "Otherwise, the courier's delivery rate decreases," says Leister.
So the courier's level of performance depends on the architecture of the thylakoids. Both the tower blocks and the walkways between them are congested. Not only the membranous structures, but the aqueous phase that surrounds them, is densely packed with proteins and small molecules. "And that reduces the mobility of the couriers significantly," Leister notes.
Earlier work done by his research group had shown that the architecture of the thylakoids has a crucial impact on the efficiency of photosynthesis under fluctuating light intensities. "When light levels are low, thylakoid membranes have to be stacked in order to maintain the efficiency of photosynthesis, and only land plants can perform this trick," he says. "Furthermore, to make things easier for the couriers, the width of the individual tower blocks must be restricted. These architectural specifications are observed in essentially all plants."
Nevertheless, there is still room for further optimization of the plastocyanin system, Leister believes. The primary weakness lies in the courier's relatively short range. "There are other theoretical solutions, which evolution has not yet explored, and these need not necessarily be less effective than those that natural selection has found," says Leister. "We are planning to test these possibilities as part of our synthetic-biology approach to crop-plant improvement."
More information: Ricarda Höhner et al. Plastocyanin is the long-range electron carrier between photosystem II and photosystem I in plants, Proceedings of the National Academy of Sciences (2020). DOI: 10.1073/pnas.2005832117
Journal information: Proceedings of the National Academy of Sciences
Provided by Ludwig Maximilian University of Munich