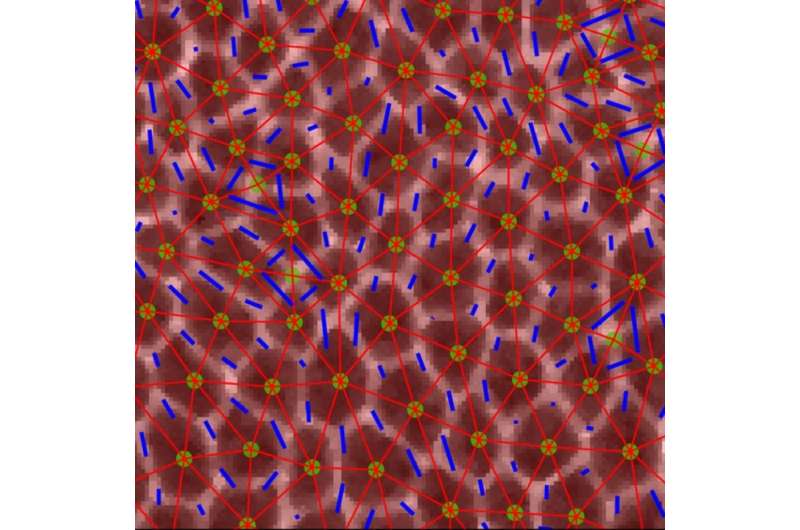
Confocal image of epithelial cells during dramatic tissue flows in the Drosophila embryo. Images of cells with overlaid triangles that were used to quantify anisotropy in the tissue. Credit: Xun Wang & Matthias Merkel/Columbia Engineering & Syracuse University
As embryos develop, tissues flow and reorganize dramatically on timescales as brief as minutes. This reorganization includes epithelial tissues that cover outer surfaces and inner linings of organs and blood vessels. As the embryo develops, these tissues often narrow along one axis and extend along a perpendicular axis through cellular movement caused by external or internal forces acting differently along various directions in the tissue (anisotropies). Researchers have long wondered how simple clusters of cells inside developing embryos transform into tissues and organs—how do tissues physically change shape in the embryo? Might they turn from "solids" into "fluids" at specific times in development to make it easier to rapidly sculpt functional tissues and organs?
Watching and measuring what happens in tissues inside the human embryo is currently not possible, and it is still very difficult to do this in mammalian models like mice. Because humans and the fruit fly Drosophila share so many biological similarities, researchers from Columbia Engineering and Syracuse University decided to tackle this problem by focusing on fruit flies. In a paper published online May 29 in PNAS, the team reports that they can predict when the tissue will begin to rapidly flow just by looking at cell shapes in the tissue.
"Thanks to earlier theoretical work from our colleagues at Syracuse, we thought we might be able to learn something about whether the embryonic tissues are solid or fluid by just looking at the shapes of cells in the tissue," says the study's lead PI Karen Kasza, Clare Boothe Luce Assistant Professor of Mechanical Engineering. "So we decided to try this in the fly. We're really excited about our results, which could reveal fundamental mechanisms underlying human development and point to where things can go wrong, causing birth defects."
The challenge was how to apply traditional engineering approaches to measure the mechanical properties of cells and tissues inside the flies' tiny embryos to see which tissues behave like solids, maintaining their shape and resisting flow, and which tissues behave like fluids, flowing easily and changing shape. The researchers used high-resolution confocal fluorescence imaging to take movies of embryonic development in which they could see in great detail the shape and packings of cells in tissues inside the fly embryo. They focused on a very fast-moving developmental event in which the embryonic tissue rapidly changes shape to elongate the head-to-tail body axis of the fly (something that also happens in most animal embryos).
Movie of epithelial cells during dramatic tissue flows, which elongate the head-to-tail body axis of the Drosophila embryo. Credit: Xun Wang/Columbia Engineering
By combining experimental studies in the fruit fly embryo at Columbia with theoretical modeling approaches at Syracuse, the researchers demonstrated that the shapes and alignment of cells within tissues can help to both explain and predict how tissues change shape during development and how defects in these processes can result in abnormalities in embryo shape. "It was a fantastic collaboration between experiment and theory," Kasza observes.
Adds Lisa Manning, co-author of the study and William R. Kenan, Jr. Professor of Physics at Syracuse, "From the theory side, it was really unclear what collective mechanisms allow the cells to easily rearrange during tissue elongation. With Professor Kasza's group, who has some of the best tools in the world to study mechanical properties of fruit fly tissue, we were really able to nail down precisely how changes to cell shapes drive changes to tissue mechanics. It is amazing that we can now just look at a snapshot of cell shapes in the fruit fly and predict how cells will move, with no fit parameters."
A surprise for the researchers was that they could anticipate when the tissue would begin to flow by looking at cell shapes in the tissue without any adjustable parameters in the theoretical model. But, unlike previous studies and predictions, they needed to include a new parameter—anisotropy—that described the alignment of cells within the tissue because the forces acting on and in the tissue were highly anisotropic, or varied along different directions in the tissue. What they found particularly interesting was that their findings suggest that embryonic tissue seems to become more fluid-like just before the onset of the rapid tissue flows during body axis elongation.
"This is really exciting" says Kasza, "because it suggests that the mechanical properties of the cells might be regulated biologically during embryonic development, i.e. in the genetic instructions encoded in DNA, to make it easier for tissues to change shape dramatically during brief time windows during development. This adds to a growing body of research revealing that mechanics is really crucial to understanding life."
The team is now looking at how the instructions for changes in tissue fluidity are genetically encoded. They are also exploring the mechanical properties of tissues to build quantitative models of tissue morphogenesis that will enable them to predict, design, build, and control tissue shape and tissue movements, both in developing embryos and in engineered tissues in the lab.
More information: Xun Wang et al. Anisotropy links cell shapes to tissue flow during convergent extension, Proceedings of the National Academy of Sciences (2020). DOI: 10.1073/pnas.1916418117
Journal information: Proceedings of the National Academy of Sciences