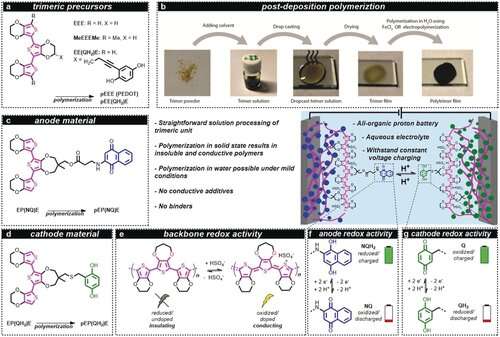
Schematic representation of the all‐organic battery concept, chemical structures/naming and polymerization method. The trimeric precursors (a) were used in the post‐deposition polymerization procedure (b) to form polymers with similar characteristics to those formed from monomeric units. In post‐deposition polymerization, the trimer is first dissolved in an organic electrolyte, followed by drop casting and drying. Subsequently, the trimer film is oxidized, either i) electrochemically in an aqueous 0.5 m H2SO4 solution by cyclic voltammetry between 0.0 and 1.21 V vs. SHE at 10 mV s−1 or by applying a potential of 0.81 V vs. SHE for 3000 s or ii) chemically by immersion into an acidic aqueous solution containing 1 m FeCl3 as oxidant, resulting in the formation of a black polymer layer. The anode material (c) consisted of pEP(NQ)E, which was formed by oxidative polymerization of EP(NQ)E. Similarly, the cathode material pEP(QH2)E (d) was formed from EP(QH2)E. Conductivity was achieved from a polythiophene backbone (e) that was oxidized/doped, for example, with HSO4−. The battery (middle) was assembled as an all‐organic proton battery using 0.5 m H2SO4 (aq) electrolyte, which enabled a rocking‐chair motion of the protons. The anode and cathode redox activity relies on the two‐electron two‐proton (2e2 H) redox process of the pendants (f and g). When the battery is charged, the quinone pendant groups are in the Q and NQH2 states, for the positive electrode (cathode) and negative electrode (anode), respectively. During discharge, the active cathode material is converted to QH2 while the anode is converted to NQ. E=3,4‐ethylenedioxythiophene; NQ=naphthoquinone; NQH2=naphthohydroquinone; P=3,4‐propylenedioxythiophene; p=polymerized; Q=benzoquinone; QH2=hydroquinone. Angewandte Chemie International Edition (2020). DOI: 10.1002/anie.202001191
Sustainable energy storage is in great demand. Researchers at Uppsala University have therefore developed an all-organic proton battery that can be charged in a matter of seconds. The battery can be charged and discharged over 500 times without any significant loss of capacity. Their work has been published in the scientific journal Angewandte Chemie.
The researchers have been able to demonstrate that their battery can be easily charged using a solar cell. Charging can also be accomplished without the aid of the advanced electronics that, for example, lithium batteries require. Another advantage of the battery is that it is unaffected by ambient temperature.
"I'm sure that many people are aware that the performance of standard batteries declines at low temperatures. We have demonstrated that this organic proton battery retains properties such as capacity down to as low as -24°C," says Christian Strietzel of Uppsala University's Department of Materials Science and Engineering.
A great many of the batteries manufactured today have a major environmental impact, not least due to the mining of the metals used in them.
"The point of departure for our research has therefore been to develop a battery built from elements commonly found in nature and that can be used to create organic battery materials," explains Christian Strietzel.
For this reason, the research team has chosen quinones as the active material in their battery. These organic carbon compounds are plentiful in nature, among other things occurring in photosynthesis. The characteristic of quinones that researchers have utilised is their ability to absorb or emit hydrogen ions, which of course only contain protons, during charging and discharging.
An acidic aqueous solution has been used as an electrolyte, the vital component that transports ions inside the battery. As well as being environmentally friendly, this also provides a safe battery free from the hazard of explosion or fire.
"There remains a great deal of further development to be done on the battery before it becomes a household item; however, the proton battery we have developed is a large stride towards being able to manufacture sustainable organic batteries in future," says Christian Strietzel.
More information: Christian Strietzel et al. An aqueous conducting redox polymer based proton battery that can withstand rapid constant‐voltage charging and sub‐zero temperatures, Angewandte Chemie International Edition (2020). DOI: 10.1002/anie.202001191
Journal information: Angewandte Chemie , Angewandte Chemie International Edition
Provided by Uppsala University