How light can a foldable and long-lasting battery be?

With the launch of wearable devices and smartphones that require a high capacity of electricity such as foldable phones and 5G phones, the interest in batteries is increasing and various battery types are under development. For example, flexible batteries embedded in a mobile watch band or wireless power sharing batteries that have developed from wireless charging. However, there is currently no manufacturing process for a battery that produces thousands of milliamp Hour (mAh) with the capacity to be foldable. Recently, a research team from Korea developed a monolithic electrode that can replace heavy copper collectors and enabled development of such a flexible battery with high capacity.
Professor Soojin Park of the Division of Chemistry and Advanced Materials Science with his postdoctoral researcher, Jaegeon Ryu and his Ph.D. student, Jieun Kang successfully developed a flexible battery with thin and three-dimensional organic electrodes in collaboration with the Korea Institute of Materials Science.
Furthermore, they were able to lower the weight of a battery by 10 times more than the conventional copper collector by using a three-dimensional copper collector. Instead of using a graphite anode, they utilized organic materials and were able to increase the energy density of a battery by four times and more. Their research was published in the recent issue of ACS Nano.
Electrical conductivity of an organic material is low and there was no solution to integrate a collector and organic material. For this reason, it had not been possible to demonstrate a monolithic electrode with organic materials before their study. The research team studied a new way to replace a current collector that makes a battery heavy and a graphite anode with low energy density in order to innovatively lower the weight of the battery.
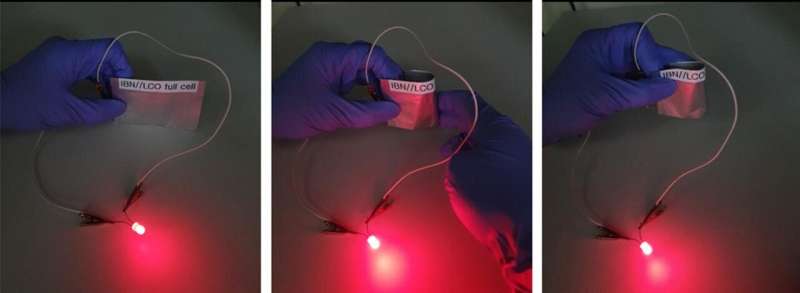
The team produced a three-dimensional structure with high electrical conductivity by using single-walled carbon nanotube (SWCNT) aerogels. Here, they constructed thin monolithic organic electrodes by coating a nanometer-scale imide-based network (IBN)2) organic material.
The three-dimensional monolithic electrodes coated with 8 nm thin and adjustable thick organic IBN layers delivered capacity of up to 1550 mA h g-1 and had the capacity to recharge more than 800 times. These electrodes were coated with organic materials. Despite their poor intrinsic electrical conductivity, they had high electrical conductivity and they also demonstrated improved electrochemical performance of the rechargeable battery by helping fast transfer of lithium through abundant redox-active sites. Moreover, thickness of the coated organic materials can be controlled easily and they were able to improve the current density of the organic electrode greatly.
The newly developed electrode can replace metal-based collectors and this enables development of a light and flexible rechargeable battery which later can be applied to wearable electronic devices, flexible devices, telecommunication and electronic vehicles of the next generation.
Professor Soojin Park who led the research commented, "We can lower the weight of a rechargeable battery enormously by using this newly developed monolithic electrode with SWCNT organic materials. This can overcome the limitations of the conventional rechargeable battery and can realize flexibility and weight lightening of an organic battery."
More information: Jaegeon Ryu et al, Three-Dimensional Monolithic Organic Battery Electrodes, ACS Nano (2019). DOI: 10.1021/acsnano.9b07807
Journal information: ACS Nano
Provided by Pohang University of Science & Technology (POSTECH)





















