Credit: CC0 Public Domain
In the six years since its inception, CRISPR gene editing has experienced ups and downs, from giddy excitement over the technology's potential to cure genetic diseases to patent disputes, ethical considerations and cancer scares. Despite recent setbacks, companies developing CRISPR therapies are forging ahead, reports an article in Chemical & Engineering News (C&EN), the weekly news magazine of the American Chemical Society.
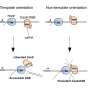
The CRISPR process makes double-stranded breaks at particular sites in DNA, specified by a guide RNA, with an enzyme called Cas9. The cell machinery then repairs the DNA break, while a CRISPR template DNA sequence introduces edits to restore a defective gene's function. The first clinical trials of CRISPR will take place within the next year. However, since May 2017, a few studies in mice and human cell lines have raised safety concerns over the technology, writes Assistant Editor Ryan Cross.
Last month, a pair of papers in Nature Medicine indicated that CRISPR doesn't work in two human cell lines unless a protein called p53 is broken or missing. Because cancer cells often have defective p53, some media coverage linked CRISPR to cancer. However, representatives from companies developing the technology downplay these results because they are not using these cell lines for their therapies. They also note that an earlier report suggesting that CRISPR is not as precise as intended has been retracted because the alleged off-target effects of the technology were later attributed to natural genetic variation in the studied mice.
More information: "Untangling the CRISPR cancer scare," cen.acs.org/business/Untanglin … -cancer-scare/96/i27
Journal information: Nature Medicine
Provided by American Chemical Society