Spinal cord injuries can be devastating because the damaged nerves do not regenerate on their own, which often leads to permanent impairment. Scientists have been investigating methods to encourage regrowth, but so far there are no treatments that reliably restore nerve function. Now, a group reports in ACS Biomaterials Science & Engineering a strategy that regrows nerve cells and restores motor function in rats with spinal cord injuries.
The current thinking is that if nerves could be encouraged to regrow past the site of a spinal cord injury, permanent disability could be prevented, according to the researchers. One possible strategy involves implanting biomaterial scaffolds at the injury site that could support new tissue growth. However, these scaffolds often fail to direct nerve regrowth past the location of the injury because it is difficult to control the direction of nerve growth, and scars can form around the implant, blocking the nerves. Thus, Sing Yian Chew and colleagues set out to design a new type of scaffold that could help nerve cells regrow and restore function.
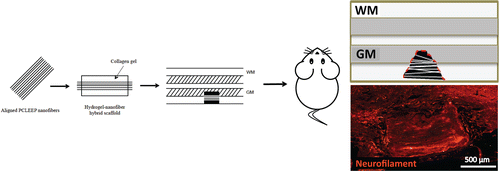
The group combined a hydrogel with aligned nanofibers to create a scaffold with a 3-D structure that can be implanted at the injury site. They also tested whether adding neurotrophin-3, a protein that encourages nerve growth, to the scaffold would help. After three months in the spinal cords of injured rats, the scaffolds—especially those laced with neurotrophin-3—had successfully encouraged nerve growth without inciting inflammation or scarring. Rats with the neurotrophin-3 scaffolds regained more motor function than those without treatment. The researchers say that these findings show that their scaffold design could someday serve as a guide for nerve regeneration techniques to treat spinal cord injuries in humans.
More information: Ulla Milbreta et al. Three-Dimensional Nanofiber Hybrid Scaffold Directs and Enhances Axonal Regeneration after Spinal Cord Injury, ACS Biomaterials Science & Engineering (2016). DOI: 10.1021/acsbiomaterials.6b00248
Abstract
Spinal cord injuries (SCIs) are followed by a complex series of events that contribute to the failure of regeneration. To date, there is no robust treatment that can restore the injury-induced loss of function. Since damaged spinal axons do not spontaneously regenerate in their native inhibitory microenvironment, a combined application of biomaterials and neurotrophic factors that induce nerve regeneration emerges as an attractive treatment for SCIs. In this study, we report the novel use of a three-dimensional (3D) hybrid scaffold to provide contact guidance for regrowth of axons in vivo. The scaffold comprises 3D aligned sparsely distributed poly(ε-caprolactone-co-ethyl ethylene phosphate) nanofibers that are supported and dispersed within a collagen hydrogel. Neurotrophin-3 was incorporated into the scaffold as an additional biochemical signal. To evaluate the efficacy of the scaffold in supporting nerve regeneration after SCIs, the construct was implanted into an incision injury, which was created at level C5 in the rat spinal cord. After 3 months of implantation, scaffolds with NT-3 incorporation showed the highest average neurite length (391.9 ± 12.9 μm, p ≤ 0.001) as compared to all the other experimental groups. In addition, these regenerated axons formed along the direction of the aligned nanofibers, regardless of their orientation. Moreover, the presence of the hybrid scaffolds did not affect tissue scarring and inflammatory reaction. Taken together, these findings demonstrate that our scaffold design can serve as a potential platform to support axonal regeneration following SCIs.
Provided by American Chemical Society