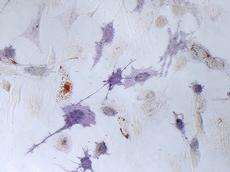
Mesenchymal Stem Cells (hMSCs) cultured on a Polyacrylamide gel for 7 days: Cells stained in blue are ALP positive which is a marker for osteogenic differentiation, while the cells that contain red oil droplets underwent adipogenic differentiation. Credit: Bojun Li and Prof. Viola Vogel / ETH Zurich
(Phys.org) -- Researchers were positive: a substrate’s softness influences the behaviour of stem cells in culture. Now other researchers have made a new discovery: the number of anchoring points to which the cells can adhere is pivotal.
How stem cells differentiate is evidently not so much a question of the stiffness of the substrate upon which they thrive, as the cells’ mechanical anchoring on the substrate surface. This is shown in a study recently published in Nature Materials by researchers from various European universities, including ETH Zurich.
Since 2006 the research community has been convinced that stem cells can “feel” the softness of materials they grow upon. Scientists mainly drew this conclusion from correlations between the softness of the substrate and the cells’ behavior.
The new research project, to which ETH-Zurich professor Viola Vogel and her doctoral student Bojun Li made a key contribution, has come to another conclusion. It reveals that the properties of the network structure of polymers are instrumental in regulating the anchoring of the collagen proteins to which the cells ultimately adhere. And these anchors influence the differentiation of stem cells.
Good protein adhesion makes surface seem “stiff”
In a series of experiments, which Britta Trappmann from Cambridge University partly conducted at ETH Zurich, the cells were applied to two different polymers of the same softness. However, the polymers differed in terms of their surface structure, which regulates the number of firmly anchored collagen proteins.
If the researchers reduced the number of well-anchored proteins on a hard surface, the cells behaved in the same way as on a soft base. If the anchors were close together, the stem cells differentiated into bone cells. If the anchors were further apart, they became fat cells. “The simple correlation that a material’s stiffness or elasticity can govern the differentiation of stem cells is therefore not universally valid”, says Vogel.
Paradigm shift in cultivation of stem cells
With their experiment, the researchers shake a paradigm. In a study conducted in 2006, scientists revealed a connection between polymer stiffness and the degree of cell differentiation. However, the researchers varied the stiffness of the polymer by varying its network structure.
The new study shows that it is this network structure, not the stiffness, that regulates how the extracellular matrix proteins adhere to the polymer. The new findings also reveal that the mechanical feedback via the extracellular collagen matrix plays a key role in cell differentiation.
Exemplary collaboration between European labs
For the study, doctoral student Britta Trappmann from Cambridge University collaborated with numerous European research teams, each of which had their own specific expertise that all came together in the project like the pieces of a mosaic. “The project needed different techniques for it to work,” says Vogel. The project was initiated by professors Fiona Watt and Wilhelm Huck in Cambridge and Nijmegen and every lab involved, including that of Prof. Joachim Spatz at the MPI in Stuttgart, made a considerable contribution towards its success with its knowhow.
Old problem considered in new light
“The findings will have a major impact on the way materials for the cultivation of stem cells are designed,” the ETH-Zurich professor is convinced. According to the scientist, it is evident that the mechanical properties of the interfaces between cells and the extracellular matrix differ on different substrates. “That has been overlooked until now,” she says. “We will differently consider how to optimize the properties of carrier media in future.” Polymeric carrier media are needed in labs and hospitals as biomedical implants, and to cultivate stem cells for the production of tissue scaffolds.
More information: Trappmann B, et al. Extracellular-matrix tethering regulates stem-cell fate. Nature Materials (2012) Published online 27 May 2012. doi:10.1038/nmat3339
Journal information: Nature Materials
Provided by ETH Zurich