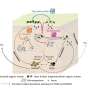
A research team has identified epigenetic signatures, markers on DNA that control transient changes in gene expression, within reprogrammed skin cells. These signatures can predict the expression of a wound-healing protein in reprogrammed skin cells or induced pluripotent stem cells (iPSCs), cells that take on embryonic stem cell properties. Understanding how the expression of the protein is controlled brings us one step closer to developing personalized tissue regeneration strategies using stem cells from a patient, instead of using human embryonic stem cells. The study was published in the Journal of Cell Science.
When skin cells are reprogrammed, many of their cellular properties are recalibrated as they aquire stem cell properties and then are induced to become skin cells again. In order for these "induced" stem cells to be viable in treatment for humans (tissue regeneration, personalized wound healing therapies, etc.), researchers need to understand how they retain or even improve their characteristics after they are reprogrammed.
Since the initial discovery of reprogramming, scientists have struggled with the unpredictability of the cells due to the many changes that occur during the reprogramming process. Classifying specific epigenetic signatures, as this study did, allows researchers to anticipate ways to produce cell types with optimal properties for tissue repair while minimizing unintended cellular abnormalities.
The researchers used reprogrammed cells to generate three-dimensional connective tissue that mimics an in vivo wound repair environment. To verify the role of the protein (PDGFRbeta) in tissue regeneration and maintenance, the team blocked its cellular expression, which impaired the cells' ability to build tissue.
"We determined that successful tissue generation is associated with the expression of PDGFRbeta. Theoretically, by identifying the epigenetic signatures that indicate its expression, we can determine the reprogrammed cells' potential for maintaining normal cellular characteristics throughout development," said first author Kyle Hewitt, PhD, a graduate of the cell, molecular & developmental biology program at the Sackler School of Graduate Biomedical Sciences, and postdoctoral associate in the Garlick laboratory at Tufts University School of Dental Medicine (TUSDM).
"The ability to generate patient-specific cells from the reprogrammed skin cells may allow for improved, individualized, cell-based therapies for wound healing. Potentially, these reprogrammed cells could be used as a tool for drug development, modeling of disease, and transplantation medicine without the ethical issues associated with embryonic stem cells," said senior author Jonathan Garlick, DDS, PhD, a professor in the department of oral and maxillofacial pathology and director of the division of tissue engineering and cancer biology at TUSDM.
Jonathan Garlick is also a member of the cell, molecular & developmental biology program faculty at the Sackler School and the director of the Center for Integrated Tissue Engineering (CITE) at TUSDM.
More information: Hewitt KJ, Shamis Y, Knight E, Smith A, Maione A, Alt-Holland A, Garlick JA. Journal of Cell Science. "PDGFRbeta Expression and Function in Fibroblasts Derived from Pluripotent Cells is Linked to DNA Demethylation" Published online February 17, 2012, doi: 10.1242/jcs.099192
Journal information: Journal of Cell Science
Provided by Tufts University