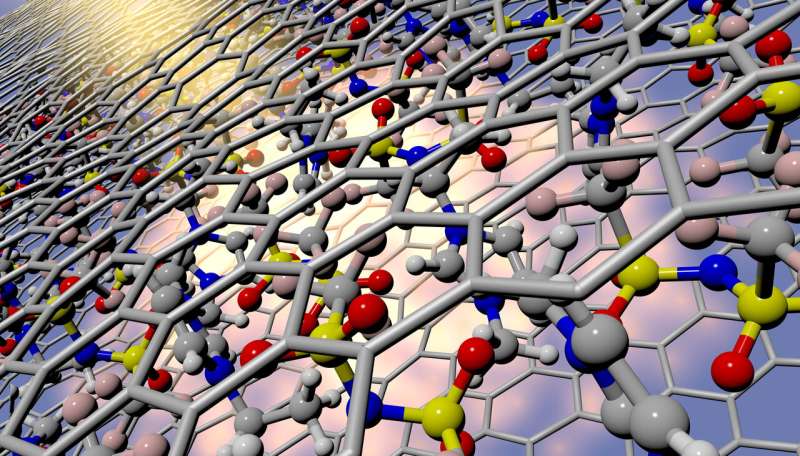
Synchrotron X-ray beam impinging upon ionic liquid molecules confined within a graphitic carbon slit-pore. X-ray scattering in the background reveals new details regarding their structure under nanoconfinement, which were validated by quantum simulations. Credit: Liam Krauss/LLNL
Room temperature ionic liquids (ILs), a special class of molten salts, promise far greater electrochemical performance compared to conventional aqueous solutions due to a suite of novel and tunable properties. Over the past two decades, ILs have been explored as a means of improving a range of different technologies, from energy storage and conversion to catalysis to electroplating of metals and semiconductors.
A prime example of where ILs can make their mark is in carbon-based supercapacitors that store electrical energy at the nanoporous electrode-electrolyte interface. How ILs assemble at this interface governs the amount of energy stored and the charging and discharging rates in devices. However, comprehensive structural insights have been slow to evolve because electrolyte behavior at interfaces and under confinement is challenging to resolve. This is especially true for ILs, which exhibit bulky, flexible and widely varying molecular configurations.
In research recently published in The Journal of Physical Chemistry Letters, Lawrence Livemore National Laboratory (LLNL) scientists coupled X-ray experiments with high-fidelity simulations to investigate a widely used family of ILs confined in carbon nanopores typically used in supercapacitors. The work represents the first study that combines first-principles molecular dynamics and X-ray scattering to analyze spatially confined ILs, enabling new insights into exotic properties that only occur within these exceptionally small spaces.
The team experimentally detected extreme disruption in the structure of the ILs, which was uniquely predicted and explained by their simulations. The team also demonstrated how deviations from typical liquid behavior depended heavily on the relative sizes of the ions and pores. Finally, despite significant deviations in structure under confinement, the study indicates that the superior electrochemical stability of ILs remains intact, which is important for maintaining the performance of energy storage devices.
"The real success is the integration between quantum-mechanical simulations, tailored nanomaterials synthesis and advanced X-ray characterization. This powerful combination of techniques offers a far more complete understanding of ILs structure in extremely narrow porous carbons," said Tuan Anh Pham, LLNL scientist in the Quantum Simulations Group and lead author on the paper. "The study represents continual efforts at LLNL in establishing interdisciplinary collaboration in the area of energy materials, like the Laboratory of Energy Applications for the Future."
LLNL researchers and co-authors on the paper, Colin Loeb and Patrick Campbell, leveraged special Lab knowledge to synthetically tune the pore sizes within high surface-area nanoporous carbon aerogels. This novel material capability enabled the team to probe with synchrotron X-rays different confined states of the ionic liquids and piece together a more comprehensive picture of the effects of confinement on structure.
For this work, LLNL forged a new collaboration with the University of Bayreuth in Germany to capitalize on key expertise in characterizing mesoscale structures.
"Interface science is such an exciting area, where we are literally just scratching the surface of an atomistic understanding of what is actually going on," said Mirijam Zobel, a faculty member in the Department of Chemistry at the University of Bayreuth and co-author on the study. "It is a rewarding experience to be part of this international team and to extend our knowledge of interfacial restructuring of complex liquids."
"I love how the different facets of our team pushed the limits of what they might be used to technically or scientifically in order to truly integrate together," said Eric Meshot, LLNL scientist and the principal investigator on the project. "We were able to uncover some key fundamental insights that have important practical implications for energy storage devices. Now we are in a unique position to be thinking more about how these insights can benefit real applications."
More information: Tuan Anh Pham et al. Structural Anomalies and Electronic Properties of an Ionic Liquid under Nanoscale Confinement, The Journal of Physical Chemistry Letters (2020). DOI: 10.1021/acs.jpclett.0c01810
Journal information: Journal of Physical Chemistry Letters
Provided by Lawrence Livermore National Laboratory