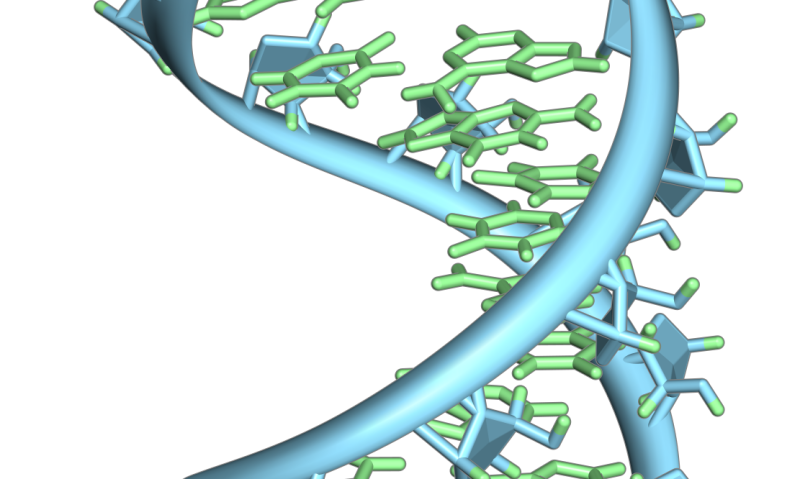
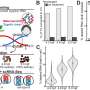
A hairpin loop from a pre-mRNA. Highlighted are the nucleobases (green) and the ribose-phosphate backbone (blue). Note that this is a single strand of RNA that folds back upon itself. Credit: Vossman/ Wikipedia
A team of researchers with the Broad Institute of Harvard and MIT has found evidence showing that using base editors can lead to unexpected RNA cellular edits. In their paper published in the journal Science Advances, the group describes their study of the CRISPR type of adenine base editor (ABE), and what they found.
ABEs convert one DNA base pair into another, allowing for the repair of mutations in some cell types without generating undesired editing effects. It is believed that ABEs also have the potential to correct almost half of all known genetic abnormalities that lead to medical disorders. The science underlying ABEs has become increasingly important to the medical community. Unfortunately, some very recent studies have found indications that ABEs might also be engaging in unexpected editing. This past March, a team found that the cytosine base editor 3 induced single-nucleotide variants at a higher-than-normal rate. And last month, another team discovered that cytosine base editors and ABEs result in off-target editing in RNA. In this new effort, the researchers sought to further test for off-target edits when using ABEs and to find a solution if it was confirmed.
In their work, the researchers analyzed the newest version ABE called ABEmax in a way that included all cellular RNA transcripts in a human cell line—and they did it with more sensitive tools than others had used. They report that they did find low levels of undesired edits in RNA samples. To get around the problem, the team developed new variant ABEs (based on inactivated wild-type Escherichia coli) that retained their ability to make the desired base edits but caused less RNA editing. They further report that the new variants were created in a way that decoupled the RNA and DNA editing process, which minimized off-target editing in both DNA and RNA.
The researchers conclude by noting that because of low levels of RNA editing and the short half-life of RNA, the degree of interference in future endeavors will likely depend on the specific applications in which they are used. They recommend that researchers seeking to minimize RNA editing use the new variant they created—they have called it ABEmaxQW.
More information: Holly A. Rees et al. Analysis and minimization of cellular RNA editing by DNA adenine base editors, Science Advances (2019). DOI: 10.1126/sciadv.aax5717
Journal information: Science Advances
© 2019 Science X Network