The new molecule linking technique can be imagined as three distinct railway wagons, each possessing two unique couplers at either end, only permitting them to be hitched in a particular order. Credit: Takeharu Haino
Manufactured polymers are ubiquitous in the market. These large molecules are used for synthetic clothing, rubbers and glues, and anything made of plastic. However, the material properties exhibited by man-made polymers rely on the sequence order taken by individual molecules comprising the polymer chain. For example, a polymer chain made up of A, B, and C molecules could potentially take the form of A-B-C-B-A or A-C-A-B-B etc. Each polymer could thus have vastly different properties.
Until now, material scientist have relied on mixing solutions, such as A, B and C together, and observing the formation of the resultant polymer, severely limiting the development of new materials. Now, Professor Takeharu Haino and Dr. Takehiro Hirao from HU's department of Chemistry have developed a way to precisely define polymer-chain order, opening up the exciting potential to design new materials.
Taking their cue from nature, where structurally well-defined biopolymers are the norm, they have developed a self-sorting strategy that regulates the order molecules take when forming long chain polymers.
Thanks to Hiroshima University researchers, we no longer need to rely on simple chemical bonds to determine polymer materiality. Credit: Takeharu Haino
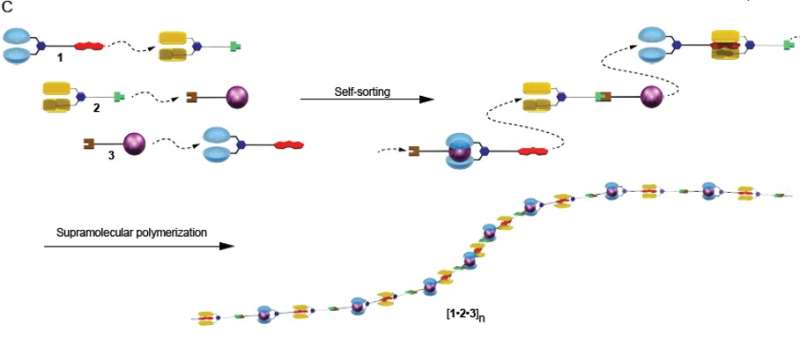
The new molecule-linking process can be imagined as three distinct train cars, each possessing two unique couplers at either end that only permit them to be hitched in a particular order. When the correct order is achieved, a train of unlimited length and complete regularity is possible.
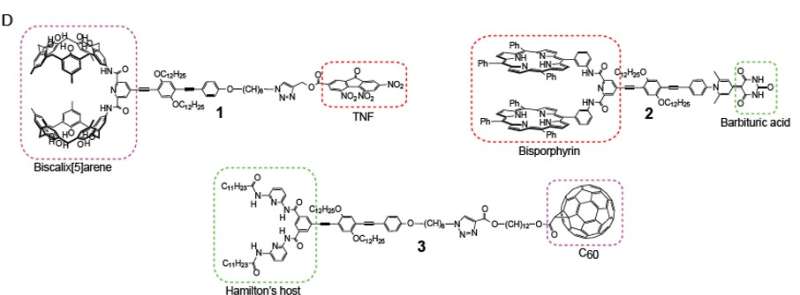
Three distinct monomer molecules were synthesized in the HU lab. Each one is different from the other,and they each possess two distinct bonding sites located at opposite ends of the molecules.
Solutions made up of these new molecules, mixed in stages, form couplet solutions. Molecule 1 bonded with molecule 2 to form a solution made up of 1-2 molecules. Molecule 2 bonded with molecule 3 forming a 2-3 solution, and molecule 3 bonded with molecule 1 to form a 3-1 solution.
When these 1-2, 2-3, and 3-1 couplet molecules were then mixed in solution, they self-sorted to form a long-chain polymer in the form of 1-2-3-1-2-3, etc, a regular polymer sequence that is predetermined and self-sorting.
Taking their cue from nature, where structurally well-defined biopolymers are the norm, HU researchers have developed a self-sorting strategy that regulates the order molecules take when forming long chain polymers. Credit: Takeharu Haino
This is a completely new way of making polymers. While previous synthetic polymers involved simple covalent bonds in which molecules share electrons to bind them together, this system uses highly specific "grabber" ends on each molecule that bond with only one type of "pin" end on another molecule.
Professor Haino says that the resulting polymer is not simply a molecule, but a molecular complex—a super-molecule. This new super-molecule production method completely and accurately predicts the makeup of the end product and can be manipulated and redesigned to give new man-made polymers with properties that could prove very useful for society.
More information: Takehiro Hirao et al, Sequence-controlled supramolecular terpolymerization directed by specific molecular recognitions, Nature Communications (2017). DOI: 10.1038/s41467-017-00683-5
Journal information: Nature Communications
Provided by Hiroshima University