Kepler 452-b is looking like a good candidate for having evolved life. Credit: NASA Ames/JPL-Caltech/T. Pyl
Are we alone in the universe? This question has been with us for thousands of years, but it is only now that science is on the cusp of providing a real answer. We now know of dozens of rocky planets orbiting stars other than our sun where, for all we know, life might exist. And soon, with the launch of the James Webb Space Telescope, we will have the first chance to peer into the atmospheres of some of these worlds.
But what should we look for? In our new study, published in Science Advances, we identify combinations of planetary temperature and light conditions sufficient to give rise to the building blocks of life.
We started with what we know. On Earth, photosynthesis – the process through which plants make energy – has transformed our atmosphere from one rich in carbon dioxide to one rich in molecular oxygen. That's because plants transform carbon dioxide and water into sugars and oxygen using sunlight.
The presence of molecular oxygen may therefore indicate the presence of life, especially if it is observed alongside methane (plants and bacteria can produce methane). If we found carbon dioxide and methane along with the complete absence of carbon monoxide, this may also be a sign of life on other planets. This is because, as far as we know, there are ways that life can release lots of methane in a carbon dioxide rich atmosphere without also making lots of carbon monoxide.
There may be other possibilities, too – scientists are looking through all possible small molecules to identify biosignatures that we haven't thought of yet.
This artist’s impression shows a sunset seen from the super-Earth Gliese 667 Cc, in its star’s habitable zone. Credit: ESO/L. Calçada, CC BY-SA
The problem with 'habitable zones'
But even if we knew exactly what to look for, where should we look? It is impossible to scan the entire cosmos for life. We have to look at individual systems, a handful at a time.
To be able to host life, an exoplanet needs to be the right distance from a star for liquid water to stably exist on its surface. The zone in which this criterion is satisfied is called the "habitable zone". If we took a vial of life and dumped it on the surface of a planet in this zone, it could survive. So these planets are a good place to start looking.
However, this does not address the question of whether life could arise there on its own. Life as we know it requires a variety of molecular structures that perform various functions within the cell. These include DNA, RNA, proteins and cell membranes, which are made up of relatively simple building blocks (lipids, nucleotides and amino acids). For a long time it was a mystery where those building blocks came from, but recently there have been major breakthroughs in determining how they arose on the surface of the early Earth.
For example, shining ultraviolet light on hydrogen cyanide (a chemical compound that exists in nature) in water, along with an negatively charged ion (an atom that has gained electrons) such as bisulfite, leads to simple sugars.
Hydrogen cyanide is abundant in the "protoplanetary disks" which form solar systems and in comets, and can be formed on a planet's surface by impact. The bisulfite on Earth probably developed from sulphur dioxide from volcanoes being absorbed into water – something that could also happen on exoplanets.
James Webb telescope. Credit: Pixabay
In certain environments, with the right conditions, hydrogen cyanide and a negatively charged ion can lead to the formation of many of life's building blocks selectively and at large concentrations. But the reactions depend on having the right amount of UV light. In the absence of light, these same molecules – hydrogen cyanide and bisulfite – slowly react to form products that do not lead to the building blocks of life.
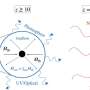
Origin of life zone
The speed of these reactions in the light and in the dark can both be measured in the lab – and that's what we did in our new study. Comparing these speeds allowed us to delineate an "abiogenesis zone" (abiogenesis means "origin of life") – the region at the right distance from a star for chemistry in the light to outcompete the chemistry in the dark.
For stars like our sun, the abiogenesis zone overlaps with the habitable zone. But for cooler stars, the story is more complicated. When cool stars are inactive, the abiogenesis zone is too close to the star to overlap with the habitable zone. But cool stars can also be very active, producing large and frequent flares. Are these flares sufficient to drive the chemistry that leads to life's building blocks? It may be possible, but much more work needs to be done to confidently identify planets around them as suitable for life.
We cross referenced our results with a catalogue of known exoplanets that are classified to be in the habitable zone to identify those that are primed for life. We found two candidates. Kepler-452b is the smallest exoplanet we know that resides definitively located in both the habitable and abiogenesis zones. Exoplanet Kepler-62e may also be in the abiogenesis zone, but it is not as likely to be rocky.
Sadly both of these exoplanets are too far away for the James Webb telescope to investigate. While we didn't find any exoplanets nearby in both the habitable and abiogenesis zones, we are discovering such worlds at breathtaking speed – with several thousands discovered already. So it may not be long until we do. For example, the Transiting Exoplanet Survey Satellite (TESS) has a chance of finding more systems like Kepler-452b that are closer to home. Until then, we could also use the method to probe moons around giant gas planets within habitable zones to find out if they are primed for life.
Although this is exciting, it should be noted that it is very difficult to solve a problem on the basis of a single data point. Right now, Earth is the only data point we have for life. In the future, if we find multiple examples of life, concepts like the abiogenesis zone can be used to test the predictions of different origin of life theories and gain new insight about how life started on Earth and whether it could have started any other way. But of course it will be amazing enough simply to discover life somewhere outside our solar system.
Provided by The Conversation
This article was originally published on The Conversation. Read the original article.![]()