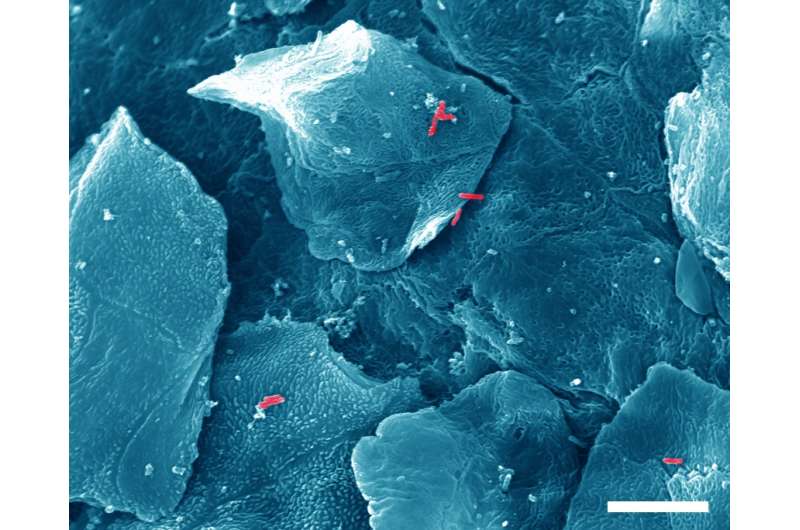
Upon infection with bacteria (red), the superficial layer of mucosal cells lifts off. Credit: P. Muenzner-Voigt and C. Hentschel
Mucous surfaces in the nose, throat, lungs, intestine, and genital tract are points of first contact for many pathogens. As a defensive strategy, most animals (and humans) can rapidly exfoliate these surfaces (i.e., shed the surface layer) to get rid of any attached attackers. A study published on May 12th in PLOS Pathogens reveals a common strategy by bacteria to prevent exfoliation and so gain extra time to colonize the mucosa or penetrate the mucosal barrier.
Christof Hauck, from the University of Konstanz, Germany, and colleagues had recently shown that one type of bacteria, Neisseria gonorrhoeae, which colonize the urogenital tract, are able to suppress exfoliation. In the process, the bacteria use proteins called Opa to bind to members of the CEACAM family of host proteins (for CEA-related cell adhesion molecules) that are expressed on mucosal cells. This binding somehow makes human cells on the surface layer of the mucosa more sticky and less likely to detach from the layers underneath.
Most pathogen-host interactions are quite complex and involve several bacterial proteins—often referred to as "virulence factors" because they enable the microbes to cause disease. In this study, the researchers examined whether engaging CEACAM is not only necessary but also sufficient to inhibit exfoliation, or whether other factors and processes are also required.
They started by genetically engineering bacteria from a normally harmless strain of E. coli (that don't have their own CEACAM binding proteins) so that they express Opa proteins. They found that this single change allowed the modified E. coli not only to bind the human CEACAM proteins but also to inhibit exfoliation.
The researchers also worked with tissue culture cells that don't have any members of the CEACAM family. If these cells are infected with Opa-containing Neisseria or E. coli strains, they do not change their adhesive properties. In contrast, if a gene expressing CEACAM is introduced into these cells, they do become sticky when infected with Opa-containing bacteria, but not when infected with the harmless E. coli strain that lacks the Opa gene.
Taken together, these results show that it is the Opa-CEACAM interaction that triggers the increased 'stickiness' of the infected cells. The experiments described so far were done in human cells cultured together with bacteria in plastic dishes, and stickiness was measured by how well the host cells stuck to the plastic surface.
To look at the interaction between bacteria and real mucosal surfaces, the researchers studied transgenic mice engineered to express high levels of human CEACAM on their mucosal surfaces. When such mice were infected intra-vaginally with Opa-expressing bacteria, the researchers found that many bacteria were sticking to the mucosal surface. They also recovered large numbers of bacteria 24 hours later, indicating that the initial ability to stick translates into subsequent successful 'colonization' of the mucosa.
In contrast, when control mice that expressed only mouse CEACAM on their mucosa were infected with Opa-containing bacteria, very few bacteria were found stuck to the vaginal mucosa. Similarly, when the mice expressing human CEACAM were infected with harmless E. coli (without Opa), few bacteria were found attached to the vaginal lining—presumably because exfoliation worked efficiently—and almost none of them were still present 24 hours later.
Several other disease-causing bacterial strains have proteins that can also bind CAECAM but are unrelated to Opa. To see whether those proteins work like Opa, the researchers tested one of the strains in the CEACAM-expressing mice. They found that large numbers of bacteria expressing Afa/Dr (the unrelated CEACAM-binding protein in question) were stuck to the vaginal wall following infection, and these bacteria were also successful in colonizing the vagina subsequently.
Based these results, the researchers propose that "CEACAM-binding adhesins have independently evolved in multiple gram-negative bacterial pathogens [...] as a means to facilitate the initial, species-specific contact with the mucosa of an appropriate host organism and to counteract the detachment of superficial cells". They conclude that "detailed mechanistic insight into this process and the ability to manipulate exfoliation might help to prevent or treat bacterial infections".
More information: PLOS Pathogens, DOI: 10.1371/journal.ppat.1005608
Journal information: PLoS Pathogens
Provided by Public Library of Science