New research could make explosives detection more sensitive. Credit: Transportation Security Administration
Scientists are reporting a simple way to improve the sensitivity of the test often used to detect traces of explosives on the hands, carry-ons and other possessions of passengers at airport security screening stations. Their report appears in ACS' The Journal of Physical Chemistry C.
Yehuda Zeiri and colleagues explain that most tests for traces of explosives begin by rubbing a swab made from glass fiber, Teflon or cotton over the suspect material. Analysis of the swab in a detector—usually a device called an ion mobility spectrometer—alerts agents to any explosive residues on the swab material. Common explosives like TNT are solids with very low vapor pressure at room temperature, so the best way to detect them is to search for particulate traces that rub off on clothing and luggage. To help security agencies prevent attacks more successfully, the researchers studied how explosive particles adhere to surfaces and how they could improve swabs to pick up even smaller amounts of explosives.
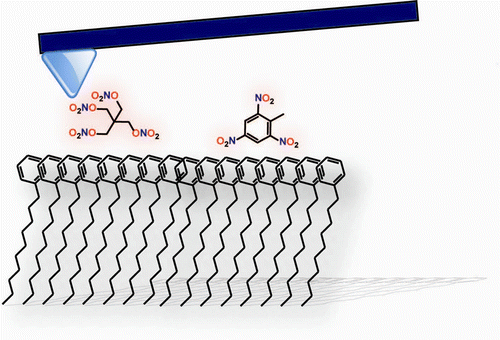
Using an atomic force microscope to measure the adhesive forces between explosive particles and different self-assembled monolayers, the scientists concluded that swab fabrics could be improved to collect smaller amounts of explosives by peppering them with hydroxyl, phenyl and amine functional groups. They believe that such additions could enhance the binding between the swab and irregularly shaped explosive particles.
More information: "Adhesion of Standard Explosive Particles to Model Surfaces" The Journal of Physical Chemistry C, 2012. DOI: 10.1021/jp303622n
Abstract
The adhesion of explosive particles to substrates with tailored chemical nature was analyzed by atomic force microscopy (AFM). Four different explosives were studied: TNT, RDX, HMX, and PETN. Two types of measurements were performed: in the first, a self-assembled monolayer (SAM) with different end groups was deposited on the tip and used to measure adhesion forces to an explosive particle surface. In the second type of experiment, the explosive particle was glued to the cantilever, and its adhesion force to a SAM-covered gold-plated glass substrate was measured. All experiments were performed both in ambient air and under water. The study shows that −OH and −C6H5 end groups lead to increased adhesion. In addition, we found that capillary forces have significantly contributed to the adhesion of explosive particles.
Journal information: Journal of Physical Chemistry C
Provided by American Chemical Society