Reducing CO2 with common elements and sunlight
An international collaborative research group including Tokyo Institute of Technology, Universite PARIS DIDEROT and CNRS has discovered that CO2 is selectively reduced to CO when a photocatalyst composed of an organic semiconductor material and an iron complex is exposed to visible light. They have made clear that it is possible to convert CO2, the major factor of global warming, into a valuable carbon resource using visible light as the energy source, even with a photocatalyst composed of only commonly occurring elements.
In recent years, technologies to reduce CO2 into a resource using metal complexes and semiconductors as photocatalysts are being developed worldwide. If this technology called artificial photosynthesis can be applied, scientists would be able to convert CO2, which is considered the major factor of global warming and is being treated as a villain, into a valuable carbon resource using sunlight as the energy source.
Complexes and inorganic semiconductors containing precious and rare metals such as ruthenium, rhenium, and tantalum have been used in highly active photocatalysts reported so far. However, considering the tremendous amount of CO2, there was a need to create new photocatalysts made only with elements widely available on Earth.
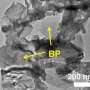
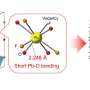
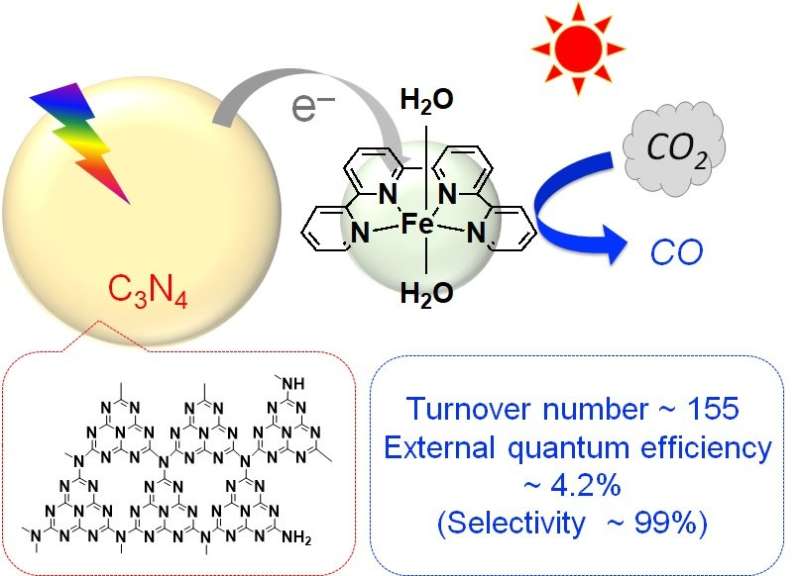
Professor Osamu Ishitani, Associate Professor Kazuhiko Maeda, and their collaborators report that by fusing carbon nitride, an organic semiconductor, with a complex made of iron and organic materials and using it as a photocatalyst, they succeeded in turning CO2 into a resource at high efficiency under the condition of exposure to visible light at ordinary temperature and pressure.
By combining the organic semiconductor carbon nitride, made of carbon and nitrogen, with an iron complex and using it as a photocatalyst, they found that they could reduce carbon dioxide (CO2) to carbon monoxide (CO) at high efficiency. This photocatalytic reaction progresses when exposed to visible light, which is the major component in the wavelength band of sunlight. The carbon nitride absorbs visible light and drives the migration of electrons from the reducing agent to the iron complex, the catalyst. The iron complex uses that electrons to reduce CO2 to CO. The turnover number, the external quantum efficiency, and the selectivity of CO2 reduction—performance indicators for the formation of CO—reached 155, 4.2%, and 99%, respectively. These values are almost the same as when precious metal or rare metal complexes are used, and about ten times more than photocatalysts reported so far using base metals or organic molecules.
This research was the first to demonstrate that CO2 can be reduced into a resource efficiently using sunlight as the energy source, even by using materials which exist abundantly on Earth, such as carbon, nitrogen, and iron. Tasks remaining are to further improve their function as a photocatalyst and to succeed in fusing them with oxidation photocatalysts which can use water, which exists abundantly on Earth and is inexpensive, as a reducing agent.
More information: Claudio Cometto et al, A Carbon Nitride/Fe Quaterpyridine Catalytic System for Photostimulated CO2-to-CO Conversion with Visible Light, Journal of the American Chemical Society (2018). DOI: 10.1021/jacs.8b04007
Journal information: Journal of the American Chemical Society
Provided by Tokyo Institute of Technology